|
 |
- Search
AbstractA 60-year-old woman suffered from recurrent femur neck fracture. Laboratory data showed serum hypophosphatemia, elevated alkaline phosphatase, normal serum calcium levels, and normal parathyroid hormone levels. Radiological examinations revealed a tumor in the right maxillary alveolar bone. The nasal cavity mass was removed, and the histological features were those of glomangiopericytoma. After removal of the tumor, some of the laboratory data normalized. Based on the clinical features, histopathological diagnosis and postoperative course of events, a diagnosis of glomangiopericytoma causing oncogenic osteomalacia was confirmed. We report a case of oncogenic osteomalacia caused by sinonasal glomangiopericytoma.
Glomangiopericytoma is a sinonasal type hemangiopericytoma that accounts for less than 0.5% of all sinonasal tumors. Histologically, this tumor is composed of very diffuse subepithelial proliferation of closely-packed spindle cells growing in various patterns. Immunohistochemical staining reveals a strong positive reaction to smooth muscle actin and a negative reaction to CD31 and CD34 [1,2].
Oncogenic osteomalacia is related to benign or malignant tumors of the bone and soft tissue. Clinically, it manifests as various symptoms characteristic of hypophosphatemic osteomalacia, such as recurrent fractures, bone pain, and gait disturbance. In recent publications, most tumors related to oncogenic osteomalacia were reported to be mesenchymal in origin, but we here report a patient with oncogenic osteomalacia caused by sinonasal glomangiopericytoma.
The case report was approved by the Samsung Medical Center Institutional Review Board.
A 60-year-old female patient had an operation on two femur neck fractures 6 years prior to presentation at another hospital. She had had no episode of familial or any other notable disorder, and so she was initially treated with medication for adult-onset osteomalacia. Even after the operation, she took analgesics for constant hip pain on both sides.
Three years prior to presentation, she fell down and visited an orthopedic clinic for excruciating left hip pain and underwent an operation for recurrent femur neck fractures. While the operation, a work-up for a pathologic fracture was performed. During the work-up, a tumor in the right maxillary sinus was discovered. Tumor resection via a Caldwell-Luc approach was conducted, but due to severe bleeding, the tumor was only partially removed. Histological examination at another hospital showed that the sinonasal tumor was a spindle cell hemangioma. At that time, the patient was informed by the rhinology surgeon that reoperation was needed.
After the orthopedic operation, her hip pain worsened on both sides. Conservative management for eight months did not do much to relieve the pain, so the patient visited our hospital for definite treatment at the Department of Orthopedic Surgery in July 2011. According to the patient's imaging work-up, the orthopedic surgeon diagnosed a previous non-union fracture site and decided to reoperate on both hip joints. Laboratory data revealed a normal serum calcium level of 8.5 mEq/dL (normal range, 8.4 to 10.2 mEq/dL), a low phosphate level of 1.4 mg/dL (normal range, 2.5 to 4.5 mg/dL), an elevated alkaline phosphatase level of 466 U/L (normal range, 42 to 98 U/L), normal serum parathyroid hormone (PTH), and normal vitamin D2 and D3. She underwent bilateral total hip replacement arthroplasty on October 11, 2011. She was referred to our Otorhinolaryngology Department for surgery of the residual tumor on the right maxillary sinus.
A residual tumor in the right maxillary sinus posterior wall was detected through paranasal sinus computed tomography (Fig. 1), and image-guided endoscopic tumor resection via a Caldwell-Luc approach was carried out on October 26, 2011. According to the operational findings, the tumor in the maxillary sinus was nearly resected, but the tumor located close to the internal maxillary artery and the masseter muscle was not completely resected due to the risk of severe complications. On histological examination, numerous thin-walled, branching staghorn vessels were surrounded by oval to spindle-shaped cells (Fig. 2A). Immunohistochemical staining confirmed that the endothelial cells stained with antibodies to CD31, CD34 (Fig. 2B, C), and smooth muscle actin (Fig. 2D). The cells were positive for smooth muscle actin and negative for CD31 and CD34. These findings were compatible with glomangiopericytoma which we concluded to be the cause of the oncogenic osteomalacia.
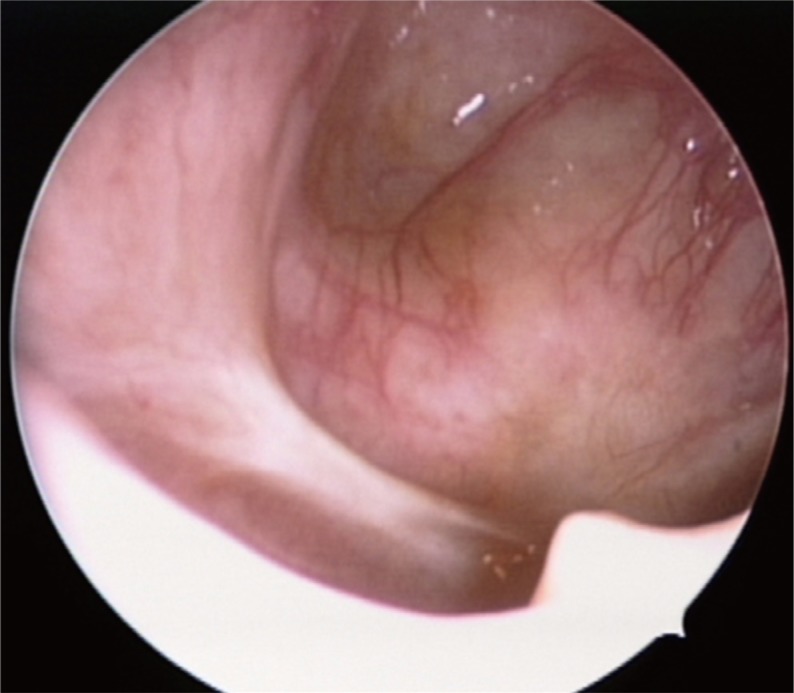
Total follow-up period of 10 months have passed by until now. Postoperative endoscopic findings through the inferior meatal antrostomy showed no evidence of residual or recurrent tumors (Fig. 3). Although it was not a complete resection, the patient's serum phosphorus levels increased from 1.4 mg/dL to 2.2 mg/dL immediately after the operation and 2.9 mg/dL 6 months after the operation (normal range, 2.5 to 4.5 mg/dL), serum calcium levels increased from 8.5 mEq/dL before the operation to 10 mEq/dL immediately after the operation and 10 mEq/dL 6 months after the operation (normal range, 8.4 to 10.2 mEq/dL), and alkaline phosphatase, which was 466 U/L before the operation, decreased to 224 U/L immediately after the operation and 113 U/L 6 months after the operation (normal range, 42 to 98 U/L).
Recurrent bone fracture and osteomalacia also have many possible causes, including trauma, oncogenic osteomalacia, auto-immune disorders, deficiency disorders such as rickets, congenital or developmental disorders. Among the causes listed, hypophosphatemia, normal or slightly low serum calcium, normal PTH, elevated alkaline phosphatase, normal 25-hydroxyvitamin D3 and low 1,25-dihydroxyvitamin D3 levels are the usual characteristics of oncogenic osteomalacia. These biochemical abnormalities are normalized by tumor removal [3].
Until now, there were 308 cases of oncogenic osteomalacia described in the world's literature over 61 years [4]. There are many kinds of histology in the tumors causing this condition. They are of mesenchymal cell line and tend to be benign. Hemangiopericytoma, angiofibromas, hemangiomas, osteoblastomas, osteosarcomas, and neurofibromas etc. have been reported. In our knowledge, sinonasal hemangiopericytomas (glomangiopericytoma) causing oncogenic osteomalacia have been reported 11 cases [5,6,7]. But, glomangiopericytoma arising from maxillary sinus causing oncogenic osteomalacia have not been reported until this case.
Majority of cases had experienced osteomalacia symptoms preceding diagnosis of a tumor. Therefore, physicians must keep in mind the possibility of oncogenic osteomalacia when hypophosphatemic osteomalacia is diagnosed.
Oncogenic osteomalacia is an acquired disease which occurs very rarely with hypophosphatemia as its major feature. Underlying diseases affecting the occurrence of hypophosphatemia need to be assessed thorough differential diagnosis. Genetic disease and acquired disease are main factors affecting hypophosphatemia. When the disease occurs at an early age, it can be classified as a genetic disease.
Autosomal-dominant hypophosphatemic rickets (ADHR), X-linked hypophosphatemia (XLH), hereditary hypophosphatemic rickets with hypercalciuria (HHRH) are examples of such genetic diseases. Most cases of XLH occur in early childhood, whereas ADHR can be seen during childhood or adulthood. Due to this important distinguishing factor, patient's medical and family history should be evaluated very carefully. Genetic disease may also accompany enamel hypoplasia, so a patient's dental record should also be carefully examined. HHRH can be distinguishable from oncogenic osteomalacia by looking into the plasma FGF23 level. Plasma FGF23 level is high in oncogenic osteomalacia and low in HHRH [8].
Underlying acquired disease of hypophosphatemia is mostly related to renal tubule damage which is caused by medications or toxins. Direct damage to the renal tubule show the same generalized tubulopathy as the genetic Fanconi-type tubulopathy. Genetic Fanconi-type tubulopathy is caused by burns, cadmium and lead exposure, aminoglycoside antibiotics, cisplatin, and tefovir. Diseases with this type of tubular damage and oncogenic osteomalacia have an important differentiating factor. As mentioned earlier, oncogenic osteomalacia shows high level of plasma FGF23, whereas tubular damage shows low level of plasma FGF23. Dietary deficiency, total parenteral nutrition, organ transplant, hematologic malignancy may also be associated with hypophosphatemia, so differentiation should be carefully approached.
Although the pathophysiology of oncogenic osteomalacia has not been specifically identified, tumor production of a humoral factor has been considered the most probable mechanism of pathogenesis up until now. Accordingly tumor inducing humoral factor inhibits 25-hydroxyvitamin D3-1-hydroxylase activity, which leads to decreases in 1,25-dihydroxyvitamin D3 synthesis [9].
Histologically, hematoxylin-eosin staining reveals a prominent pericytic vascular pattern, with thin-walled, branching vessels, often with a staghorn shape. However, many benign and malignant soft tissue tumors share this vascular pattern. Because of this, immunohistochemical staining is very important in the differential diagnosis. Glomangiopericytoma shows a typical positive reaction to actins and vimentins and a negative reaction to CD31 and CD34 on immunohistochemical staining [1].
Difficulties in the differential diagnosis are encountered with benign or borderline vascular-rich spindle cell tumors such as a solitary fibrous tumor, lobular capillary hemangioma, angiofibroma, or a leiomyoma. Solitary fibrous tumors are very rare and account for less than 1% of all sinonasal tumors. They occur more commonly in the nasal cavity than the paranasal sinus. The distinguishing feature of glomangiopericytoma lies in immunohistochemical staining, as it shows a strong positive reaction to CD34. Unlike glomangiopericytoma, lobular capillary hemangiomas show a positive reaction to CD31 and CD34 on immunohistochemical staining. Leiomyomas are very rare sinonasal tumors. A major difference between leiomyomas and glomangiopericytomas is eosinophilicity and prominent fascicular growth. Angiofibromas normally occur in male adolescents and are very rare in adults [1].
Oncogenic osteomalacia is very rare condition, and glomangiopericytom is very difficult to diagnose. Otorhinolaryngologists should keep in mind the possibility of oncogenic osteomalacia and its extremely rare cause of maxillary glomangiopericytoma.
References1. Dandekar M, McHugh JB. Sinonasal glomangiopericytoma: case report with emphasis on the differential diagnosis. Arch Pathol Lab Med. 2010 10;134(10):1444-1449. PMID: 20923298.
2. Higashi K, Nakaya K, Watanabe M, Ikeda R, Suzuki T, Oshima T, et al. Glomangiopericytoma of the nasal cavity. Auris Nasus Larynx. 2011 6;38(3):415-417. PMID: 21093999.
3. Sakamoto A, Oda Y, Nagayoshi Y, Iwakiri K, Tamiya S, Iwamoto Y, et al. Glomangiopericytoma causing oncogenic osteomalacia: a case report with immunohistochemical analysis. Arch Orthop Trauma Surg. 2001;121(1-2):104-108. PMID: 11195105.
4. Jiang Y, Xia WB, Xing XP, Silva BC, Li M, Wang O, et al. Tumor-induced osteomalacia: an important cause of adult-onset hypophosphatemic osteomalacia in China. Report of 39 cases and review of the literature. J Bone Miner Res. 2012 9;27(9):1967-1975. PMID: 22532501.
5. Gonzalez-Compta X, Manos-Pujol M, Foglia-Fernandez M, Peral E, Condom E, Claveguera T, et al. Oncogenic osteomalacia: case report and review of head and neck associated tumours. J Laryngol Otol. 1998 4;112(4):389-392. PMID: 9659507.
6. Beech TJ, Rokade A, Gittoes N, Johnson AP. A haemangiopericytoma of the ethmoid sinus causing oncogenic osteomalacia: a case report and review of the literature. Int J Oral Maxillofac Surg. 2007 10;36(10):956-958. PMID: 17498926.
7. Brandwein-Gensler M, Siegal GP. Striking pathology gold: a singular experience with daily reverberations: sinonasal hemangiopericytoma (glomangiopericytoma) and oncogenic osteomalacia. Head Neck Pathol. 2012 3;6(1):64-74. PMID: 22430770.
8. Jonsson KB, Zahradnik R, Larsson T, White KE, Sugimoto T, Imanishi Y, et al. Fibroblast growth factor 23 in oncogenic osteomalacia and X-linked hypophosphatemia. N Engl J Med. 2003 4;348(17):1656-1663. PMID: 12711740.
9. Wilkins GE, Granleese S, Hegele RG, Holden J, Anderson DW, Bondy GP. Oncogenic osteomalacia: evidence for a humoral phosphaturic factor. J Clin Endocrinol Metab. 1995 5;80(5):1628-1634. PMID: 7745010.
Fig. 1Preoperative computed tomography. Coronal (left) and axial (right) enhancing computed tomography scans showing a residual tumor with mild heterogeneous enhancement in the right maxillary sinus posterior wall (arrows). 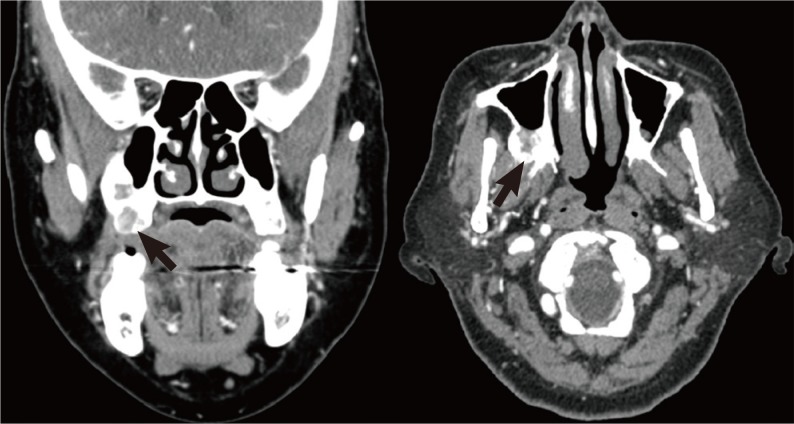
Fig. 2Pathologic findings. H&E staining (×180) revealed numerous thin-walled, branching staghorn vessels surrounded by oval to spindle-shaped cells (A). Immunohistochemical staining showed a negative reaction to CD31 (B), a negative reaction to CD34 (C), and a positive reaction to smooth muscle actin (D). (B-D) Immunohistochemistry, ×250. 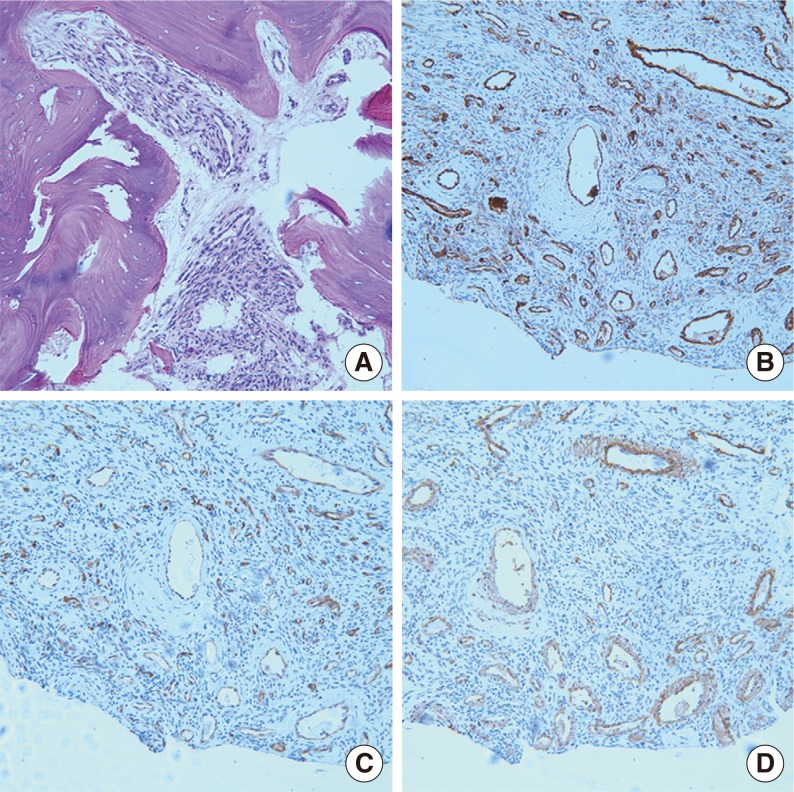
|
|
|||||||||||||||||||||||||||||||||||||||||||