Yagi: Nystagmus as a Sign of Labyrinthine Disorders-Three-Dimensional Analysis of Nystagmus-
Abstract
In order to diagnose the pathological condition of vertiginous patients, a detailed observation of nystagmus in addition to examination of body equilibrium and other neurotological tests are essential. How to precisely record the eye movements is one of the goals of the researchers and clinicians who are interested in the analysis of eye movements for a long time. For considering that, one has to think about the optimal method for recording eye movements. In this review, the author introduced a new method, that is, an analysis of vestibular induced eye movements in three-dimensions and discussed the advantages and limitations of this method.
Keywords: Nystagmus; Three-dimensional analysis; Labyrinthine disorders; Equilibrium; Vertigo
INTRODUCTION
In order to diagnose the pathological condition of vertiginous patients, a detailed observation of nystagmus in addition to examination of body equilibrium and other neurotological tests are essential. For this purpose, methods for precise observation and analysis of the eye movements have been studied and are being developed until now.
There are two major eye movements, which are the smooth pursuit and saccade. The smooth pursuit is characterized by slow eye movements consisting of 1 to 30 deg/sec and track the target, which appeared in one's view. The slow phase of nystagmus thought to be a kind of smooth pursuit. The saccade is a quite rapid eye movement, whose maximum speed reaches up to 400 to 600 deg/esc. The quick phase of nystagmus thought to be a sort of saccade.
How to precisely record the eye movements, which have these frequency characteristics mentioned-above, is one of the goals of the researchers and clinicians who are interested in the analysis of eye movements for a long time. For considering that, one has to think about the optimal method for recording eye movements. The following can be some of the points needed for an optimal method for recording eye movements.
1) can be recorded three dimensionally, horizontal, vertical, and torsional 2) can be recorded linearly up to 90 deg, except for torsional movement 3) can record fine movements, up to several seconds 4) have a wide frequency response, 0 to several hundreds Hz 5) devise for recording should not disturb the examinee's view 6) no necessity to touch the examinee's eye 7) no or less influence by the examinee's head movement
However, there is no such devise which can satisfy all the above-mentioned points at least at present.
HOW TO OBSERVE AND RECOTD THE EYE MOVEMENTS
Direct observation
The observation of the subject's eye and recording of its characteristic is rather primitive, but still most frequently used in the clinical examination. The observation method is performed under naked eye, under Frenzel glasses, and through video images. For the video image observation, the infrared CCD camera is most commonly used at present. The advantage to use this camera is that the examiner can observe eye movements in a lit room through a video monitor ( Fig. 1), since the inside of the goggle with the camera is completely dark. Also many persons, for example physicians, nurses, students, even the relatives of the patients, can simultaneously observe through a TV monitor. By this method we can more frequently detect nystagmus as compared to that under Frenzel glasses ( 1).
Cine camera
Recording the eye movement on the movie film is one of the oldest methods. To analyze eye movements precisely, the path of the light, which is the reflection from the cornea of the object's light to the examinee's eye, is followed frame by frame. This method can analyze eye movements two dimensionally, that is horizontal and vertical ( 2). The resolution of this method is 2 frames/sec and later it was improved up to 6 frames/sec ( 3), this being the limitation of this method.
Still picture
For the purpose of recording the slow eye movement such as the otolith-ocular reflex, ocular counter rolling (OCR), the still picture of 1 frame/sec to several frames/sec is used. The iris striation or vessels on the sclera are used as the landmark of the objective analysis. The landmark of the upright position is used as the primary position and the change in position of this landmark is used as the indicator of changes of the angle of the torsion of the eye ( 4). However, the analysis using this method is quite time consuming, so no body uses this method now a days.
Light reflection
There are two recording methods using the light reflex. One is the reflex from the cornea and the other is the reflex from the attachment on the eyeball. The light from the outside of the eye is reflected at the front surface of the cornea first and then makes a second reflection at the back surface of the cornea. After that the light is further reflected at the anterior surface of the lens and then makes the fourth reflection at the posterior surface of the lens. These reflections are well known as Purkinje images 1 to 4. The accuracy of the recording using this reflection is less than 0.5 deg. However, this method allowed only two-dimensional recordings, horizontal and vertical. Using this principle of reflection, several new devices have been developed until recently ( 5). The three-dimensional recording was performed using the reflection mirror attached to the contact lens by Fendar ( 6). However, the difficulty in attachment to the contact lens with this mirror to the eye and the slipping off or backlash of the lens makes this method less applicable for human subjects.
Electro-Oculography (EOG)
EOG is based on the electric potential between the cornea and the retina of the eyeball (corneo-retinal potential). The cornea is an anode relative to the retina, which shows active transportation of an ion. Thus, the eyeball is thought to be a movable battery whose cathode is directed backward. The movement of the eyeball has same meaning that the movement of a battery. Then, this potential change can be amplified and recorded from the electrodes attached to the surface of the face near the eye. This potential is about 15 to 500 micro volts. This potential is linear, if the visual angle is limited to within 30 deg ( 7). EOG is the most widely used method of recording of eye movements worldwide especially for clinical examination. However, this potential changes under various conditions, in addition to the eye movement itself. The strongest effect is the illumination. The potential comes larger in the light and smaller in the dark. Gonshor ( 8) reported that the subjects should be made to adapt to the dark for about 50 min before EOG recording. This is, however, not practical during a clinical examination. In addition to that, the weakest point of EOG is that this method can record the horizontal and vertical eye movements, but cannot record the torsional eye movement.
Cornea search coil
If the coil moves in the magnetic field, the alternating voltage is induced. This is proportional to the sine of the angle between the coil and the magnetic field ( Fig. 2). Using this principle, the eye movements in a subject who wears contact lenses with a coil sitting in the magnetic field, can be amplified and then phase-detected with respect to the magnetic field to give a signal that corresponds to movements of eye in the horizontal and vertical plane. If two search coils are used, a three dimensional recording can be made. This method was first reported by Robinson ( 9). They applied this method in an animal experiment, so that the coil was implanted in the sclera of the animal's eye ( 10). After that, the contact lens with coil was developed and thereby can be used for measurement of human eye movements ( 11). This method has great advantage from the point of view of sensitivity and accuracy for recording and analyzing the eye movements, such that the resolution was estimated to be about 6 to 15 min/deg. However, the actual accuracy seems to be rather less than what would be expected from the noise level. For clinical use, this method contains several disadvantages. Wearing the contact lens is invasive to the subjects. It causes discomfort in sensation, distortion of the cornea, dryness of cornea, elevation of the intra-ocular pressure, and so on. Therefore, in spite of the accuracy of the recording and that it can analyze three-dimensionally, this method is not used in routine clinical examination, except for some experimental trials in human subjects.
Video-oculography
Otto et al. ( 12) reported the 3D analysis method using video images of the eye. They used the contact lens with a surface marker and tracked this mark with a video image analysis. From the angle between this mark and the center of the pupil, they calculated the torsional eye movement. This means they could not solve the problem of the invasive characteristics, which the search coil method has. Therefore, this method is also not used for clinical examination. There are two main systems to record and analyze the eye movements three-dimensionally using video images. One is a system, which has been reported by Yagi and his group ( 13, 14), and another is that developed by Scherer et al. ( 15). Both methods are using video image of eye for analyzing the 3D eye movement. The lightweight CCD camera is attached to the specially designed goggle ( Fig. 3A), which can be attached to the subject's head. In the goggle, the infrared LED light illuminates the eye. In this condition, the eye movements in the complete darkness can be recorded. This is an advantage for observing and recording nystagmus in patients with labyrinthine disorders whose nystagmus can be easily identified in the complete darkness. In addition, if an examiner wishes to record the eye movements in the light, one can use the goggles, which contains the infrared reflection mirrors. Through this mirror, the vision of the subject can be kept intact and only the infrared light, which leads the image of the eye, is reflected to the infrared sensitive CCD camera ( Fig. 3B). In Yagi's ( 13, 14) system, the output signals from the infrared sensitive CCD camera are fed into the hard disk of the computer thorough and AD converter. A gray-level histogram is created from the digitized image and threshold is selected to produce binary image at the gray level of the pupil. From this binary image, the center of gravity of the pupil can be calculated. Then the position of this center of gravity is used for analyzing the horizontal and vertical eye movements. To calculate the torsional eye movement, the edge detection filter is applied and 5 to 7 iris striations, which have clearly detected the edge is chosen using a computer. By monitoring each of the position of the clear edges in relation to the center of the pupil, it is possible to calculate the angles of the torsional eye movements ( Fig. 4). From each frame of the video image the computer calculates the position of the center of the gravity of pupil and the angles between this center and several iris striations. In standard NTSC system, the sampling rate of the video image is 30 Hz. To analyze the rapid eye movement, the high-speed camera is necessary. At present, the high-speed CCD cameras, which have same size and same weights as the standard CCD camera, are available. The sampling rate of these cameras is 120 Hz ( 16) and 240 Hz. The recording charts of 3D eye movement using standard and high speed CCD camera are demonstrated in Fig. 5. Scherer et al. ( 15) used the same strategies for measuring the horizontal and vertical eye movements. In other words, he used the path of the center of gravity of the subject's pupil for tracking the two eye movements, horizontal and vertical. Their algorithm generates an annulus within the iris area, and extracts the corresponding subset of image data. The contrast profile along this annulus defines the torsional position of the iris.
ANALYSIS OF VESTIBULAR INDUCED EYE MOVEMENTS IN THREE-DIMENSIONS
Caloric nystagmus
The caloric test has been widely accepted as a means for evaluating the vestibular function. The nystagmus evoked by caloric stimulation is thought to be elicited mainly by the activities of horizontal semicircular canal. From the observation of the caloric nystagmus in rhesus monkeys, it was speculated that the superior semicircular canal is also activated during the stimulation ( 17). Also from another study it has been speculated that the caloric nystagmus is arises from the three semicircular canals, in the same animal experiment ( 18). In humans, the role of vertical canals in the generation of caloric nystagmus remains controversial. Thus, it is quite reasonable to analyze the eye movement three-dimensionally to elucidate the role of vertical canals on caloric nystagmus. Yagi et al. ( 19) analyzed the caloric nystagmus from 10 healthy subjects induced by cold water in the supine and prone position. They found, in the supine position, that the three components of nystagmus were directed towards the left (10 subjects), upward (8 subjects) or downward (2 subjects), and clockwise (10 subjects) from examiner's view. In the prone position, on the other hand, the three components were directed towards the right (10 subjects), downward (5 subjects) or upward (3 subjects), and counterclockwise (10 subjects) from the examiner's view; there was no vertical direction in two subjects. Therefore, they concluded that these findings indicate that the caloric stimulation activated the three semicircular canals simultaneously. Also they speculated that the changes in the direction of the nystagmus in the supine and prone positions could be explained, at least in part, by the nonconvective component of caloric nystagmus.
Earth Vertical Axis Rotation (EVAR)
The relationship between the anatomical and the functional planes of the horizontal canal in the rotational stimulation is not clear in detail. The relationship between these two planes is studied in 16 human subjects using 3D analysis of rotational nystagmus by Suzuki ( 20). The subjects' head are firmly fixed with the specially designed head fixed device and are given pendular rotation. In her report, the position of the head was changed 40 deg antero-flection and retro-flection from the zero deg, which is based on the line through the lateral orbital edge ant upper edge of external ear canal. The average amplitude changes of horizontal, vertical, and torsional eye movements are illustrated in Fig. 6 in relation to the head position induced by a pendular rotation. The maximum amplitude of the horizontal eye movement is in around 20 deg antero-flection of the head. The amplitude of the vertical eye movement in this range of head position is small ranging from 2 to 8 deg and no vertical eye movement is seen in some subjects. In this range of head position, the smallest amplitude is observed in the head position of about 10 deg antero-flection. The amplitude change of the torsional eye movement shows similar characteristics as that of the vertical eye movement. That is, with around 10 deg antero-flection head position the amplitude is the smallest, about 3 deg, and the largest amplitude are seen in 50 deg antero- and retro-flection of the head. From this observation, it can be said that the most sensitive functional head position to the earth vertical rotation in human is about 20 deg antero-flection in human subject. If the Reid line (Horsley-Clarke line), which is quite frequently used in the animal experiment, is used as the base line, this value is interpreted as 25 antero-flection head position of Reid line. This value is quite close to the value which is as is anatomically measured in the human dry skull ( 21). However, Fetter et al. ( 22) reported that the even the head position is changed as upright, antero- and retro-flection of 30 deg; the VOR gain is almost same.
Off Vertical Axis Rotation (OVAR)
The EVAR is one of the most common methods of stimulation for evaluating the function of the semicircular canals. On the other hand, rotation about the axis tilted from the earth vertical (OVAR), which was discovered and described almost simultaneously by Guedry ( 23) and Benson et al. ( 24) has been used to investigate the response to combined semicircular and otolith organs. The OVAR generates unique 3D eye movements, known as the modulation component (MOC) and bias component (BIC). These characteristics of the eye movements induced by OVAR are observed in rats ( 25), cats ( 26, 27), monkeys ( 28, 29) and also in human subjects ( 23, 24, 30). The MOC and BIC were clearly observed in the horizontal eye movements during OVAR in human subjects ( 31, 32). On the other hand, the presence of BIC in the vertical eye movement is controversial. In addition, these studies in human subjects are performed on the eye movements of horizontal and vertical. Then, Yagi et al. ( 32) analyzed the eye movements during OVAR three dimensionally. In their study, thirty-seven healthy human subjects are seated and rotated in a computer-controlled chair in a spherical dome, which can be tilted by hydraulic pressure up to 40 deg from the earth vertical position ( Fig. 7). Clockwise rotation at a velocity of 4 deg/sec 2 is applied until the rotation speed reached up to 60 deg/sec after which a constant velocity is maintained. Thus, one full rotation took 6 seconds. After cessation of the per-rotatory nystagmus as monitored on a TV screen, the spherical dome with the rotation chair is tilted to 30 deg at a velocity of 1 deg/sec and this position is maintained for 60 sec. During this period the eye movement is recorded on a video tape recorder and analyzed off-line. During the acceleration period mainly the horizontal nystagmus towards the right is observed. This nystagmus gradually disappears with time during the constant velocity period. However, when the orientation of the gravity is changed by tilting the spherical dome to 30 deg, the eye movements with all three components can be observed ( Fig. 8A). Between the two vertical lines drawn in Fig. 8 indicate one cycle of rotation (6 sec). These eye movements continue as long as the OVAR is maintained. The horizontal, vertical and torsional eye movements exhibit regular periodic amplitude changes (MOC). Fig. 8B shows the traces of the eye position after removing the saccades using a computer program. The eyes deviate at a velocity of 1.3 deg/sec towards the left, which has been called as BIC with the MOC mentioned above. On the other hand, no such clear eye deviation is seen in the vertical and torsional eye movements. The BIC of the horizontal, vertical, and torsional eye movements is measured after removing the saccadic eye movements. The mean and standard deviation of the BIC in the horizontal, vertical, and torsional eye movements in all subjects tested are -2.45±1.36 deg/sec, 0.05±0.89 deg/sec, and -0.06±0.28 deg/sec, respectively. The BIC in the horizontal is clearly bigger than that in the vertical and torsional movements. The direction of the BIC in the vertical and torsional components shows no clear tendency deviating towards one direction, such as upward or clockwise. The MOC of three eye movements are also measured. The mean maximum amplitude and standard deviation of horizontal, vertical, and torsional eye movements are 3.93± 1.55 deg, 2.7±0.98 deg, and 4.47±1.8 deg, respectively. The amplitude of the vertical eye movement is significantly smaller than that of the horizontal and torsional eye movements. The phase of the MOC in relation to the head position is measured and is shows an example of horizontal, vertical, and torsional eye movements in relation to the LED position of fitting with least square sinusoids in Fig. 9. The average and standard deviation of the phase lag of horizontal, vertical, and torsional eye movements with respect to the origin are 270.6±48.5, 284.4±73.9, 11.6±22.2 deg, respectively. The phase of the torsional eye movement is very consistent with a small variation between each subject. From these results, in human subjects, there should be some differences in the dynamic function of the otolith system compared to that observed in monkeys ( 28). In addition to the above-mentioned study, their group did a 3D analysis during OVAR in patients with unilateral labyrinthine loss ( 33). The MOC of these patients is not different from that of normal subjects. However, the horizontal MOC is significantly smaller when the rotation is directed towards the diseased side as opposed to the healthy side. With rotation to the diseased side, most subjects exhibit a horizontal BIC to the opposite side as compared with the diseased side in normal subjects. The vertical and torsional BIC are also influenced some extent by lack of input from the unilateral labyrinth. These results indicate that a unilateral otolith organ play a major role in the production of horizontal BIC contralateral to the direction of rotation, which is strongly related to the velocity storage mechanisms in the central nervous system ( 34- 36). The above-mentioned OVAR is induced by a constant velocity trapezoid. However, there is another mode of rotational stimulation; sinusoidal rotation. Using the sinusoidal stimulation, one can simultaneously evaluate the function both the otolith organs and the semicircular canals ( 37).
THREE-DIMENSIONAL ANALYSIS OF NYSTAGMUS IN THE LABYRINTHINE DISORDERS
Significance of 3D analysis
The stimulation of individual semicircular canals elicits eye movements in planes parallel to the plane of the canal. This physiological rule has been investigated and clarified using animal experiments of many species, such as pigeons, rabbits, cats, and monkeys ( 38, 39). If it is also true in humans, the analysis of slow phase of nystagmus, whose origin is in the vestibular labyrinth, in the peripheral disorders lead to determine the pathological localization of the disease. The rotation axis of the nystagmus can be determined from the three-dimensional analysis. The calculated rotation axis of the nystagmus is then compared to the anatomical axis of each semicircular canal ( 22, 40). The rotation axis is calculated as follows. The velocity of the slow phase of each nystagmus, torsional, vertical, and horizontal, is measured from the printed-out chart of spontaneous or evoked nystagmus in a subject. From this data, the planar equation (ax + by + cz + d = 0) is applied, where, x, y, and z express the axis of torsional, vertical, and horizontal eye movements, respectively. Then the coefficient of the equation (a, b, c) in the formula is estimated.
Pressure nystagmus in patients with lateral semicircular canal
The stimulation of individual semicircular canals elicits eye movements in planes parallel to the plane of the canal as mentioned above. In humans, it is almost impossible to prove these physiological finding directly, because stimulating an individual semicircular canal or ampullary nerve as is done in animalexperiments cannot be performed in human subjects. The labyrinthine fistula is a naturally occurring pathological condition, which can be a good model to investigate the human semicircular canal - ocular reflex. Using this model, Yagi et al. ( 41) showed the fact that the human VOR also followed the above-mentioned rule as has been proved in many animal species. That is the slow phase of nystagmus, which is evoked by applying the positive pressure, is calculated. The average velocity vectors from patients with lateral canal fistula are demonstrated in Fig. 10. In this figure, in addition to the 5 patients in that report ( 41), the data from another 6 patients with lateral canal fistula are added. The velocity vectors from all subjects are well aligned with the anatomical axis of the lateral canal ( 22). These results also show no exceptions regarding the direction of the velocity vectors of the nystagmus in all eleven cases.
Nystagmus in patients with lateral canal BPPV
Horizontal canal BPPV (HC-BPPV) is more recently proposed than posterior canal BPPV (PC-BPPV) as a new entity of the positional vertigo syndrome ( 42- 44). In HC-BPPV, the vertigo is more intense as compared to that of PC-BPPV. Positional nystagmus testing shows a long lasting (more than 30 sec), direction changing (geotropic or apogeotropic) positional nystagmus with a short latency. It occurs with a change of head position to either side, but it is always stronger on one side. The vertigo is principally caused by rotation of the head in the supine position, and the nystagmus is horizontal instead of torsional-vertical. McClure ( 42) speculated that this variety of BPPV resulted from accumulation of debris in the long arm of the horizontal canal analogous to the mechanism for PC-BPPV caused by the posterior canal lesion. This speculation mainly arises from the findings of positional nystagmus, which is horizontal in nature, and the provocation head position, which mainly stimulates the horizontal canal, leading to vertigo. The longer duration of the HC-BPPV may be explained by the longer time constant of the horizontal vestibulo-ocular reflex (VOR) compared to the vertical VOR ( 45). Several researchers tried to describe the reason for the short latency of the attack and the lack of fatigability of this disease; but there are no clear explanations as yet. Also, there is no clear evidence indicating that the pathological localization of HC-BPPV is in the horizontal semicircular canal. Regarding the HC-BPPV, Yagi et al. ( 46) investigated the axes of provoked nystagmus to clarify the pathological origin of this disease using video-oculographic technique. They analyzed nystagmus in 11 patients, who were diagnosed as the HC-BPPV from their history and positional nystagmus testing. In seven patients, the velocity vectors of the slow phase of nystagmus were well aligned with the axis of the horizontal semicircular canal and in four patients were not. They found that the pathology of HC-BPPV in most patients with apogeotropic positional nystagmus has been considered to be localized in the horizontal canal. On the other hand, the velocity vectors of nystagmus in three out of four patients with geotropic nystagmus do not align with the horizontal canal axis. Based on the results of the vector analysis of the axis, a possible combination of pathologies localized in two or three semicircular canals cannot be the case in these patients. They stated that their results strongly suggest that in the patients with geotropic positional nystagmus the pathology is localized in the utricle.
Nystagmus in patients with posterior canal BPPV (PC-BPPV)
Barany ( 47) described the basic features of BPPV. He wrote, "My assistant, Dr. Carlefors, first noticed that the attacks only appeared when she lay on her right side. When she did this, there appeared a strong rotatory nystagmus to the right with an upward component. The attack lasted for about 30 sec and was accompanied by violent attacks of vertigo and nausea. If, immediately after the cessation of the symptoms, the head was again turned to the right, no attack occurred and in order to evoke a new attack in this way, the patient had to lie for some time on her back or on the left side." Barany ( 47) carried out certain further observations, and demonstrated that the factor precipitating the vertigo was not a head movement but the head position in space, and for this reason he attributed the condition to a disorder of the otolith organs. However, it was not until 1952 that Dix and Hallpike ( 48) described the provocative positioning nystagmus test maneuver and clearly defined the syndrome as "positional nystagmus of benign paroxysmal type." They came to a similar conclusion that this entity of disease originates from pathology in the otolith organ, after reviewing the clinical features of 100 patients with BPPV. Schuknecht ( 49), however, proposed a new theory for understanding this disease. In a temporal bone study of two patients with BPPV, he found amorphous deposits on the cupula of the posterior semicircular canal on the diseased side. Based on this temporal bone study he proposed the theory of "cupulolithiasis" of the posterior semicircular canal. However, he could not explain the clinical features of BPPV, such as the limited duration of the attack and the fatigability on repeated testing, and occasional prolonged periods of remission. The latency at the onset of the positioning nystagmus was also not explained by this new theory. Barany ( 47) speculated that BPPV is caused by pathology of the otolith organ since the vertigo is induced by a change in the head position relative to the gravity and not by the head movement itself. Dix and Hallpike ( 48) suggested that the pathological localization of BPPV is in the otolith organ, especially in the utricle, because they identified unilateral utricular macula degeneration at necropsy in a typical case of BPPV. These days the posterior semicircular canal is considered to be a strong candidate as a responsible site of the pathology in the inner ear. Schknecht ( 49) proposed the theory of "cupulolithiasis" from the temporal bone studies of three patients who had been suffered from severe positional vertigo. Hall et al. ( 50) suggested the free flouting debris, "canalolithiasis" in the posterior semicircular canal. Most of the descriptions above suggest that BPPV may be caused by the posterior canal or a utricular pathology. The following descriptions are somewhat different entities of BPPV than mentioned above. Fetter et al. ( 51) analyzed nystagmus of BPPV patients. They measured the velocity vectors of slow phase of provoked nystagmus in a head hanging position in five patients with BPPV using the dual-search coil method. They found a good alignment of the axes of the slow phase of nystagmus with the anatomical axis of the posterior semicircular canal in all patients tested. Aw et al. ( 52) measured the velocity vector of the slow phase of provoked nystagmus in 44 BPPV patients also using dual-search coil method. The nystagmus was provoked by Dix-Hallpike or supine ear-down test in two-axis whole body rotator. The rotation axes in twenty-one patients clustered around the posterior canal. The rotation axes in seven patients clustered around the anterior canal and those in another seven patients clustered around the horizontal canal. In the remaining 9 patients, however, the rotation axes were located between the posterior and horizontal canal axes. They concluded that, BPPV can affect one or more of the semicircular canals. Yagi et al. ( 53) investigated the nystagmus provoked in 26 patients with PC-BPPV using the video-oculographic technique by their own recording and analysis system. They hypothesized that if the origin of the nystagmus is from the posterior canal, nystagmus in the head hanging and sitting positions during the positioning nystagmus testing should be mirror images. They calculated the rotation axis from each path of the slow phase of positioning nystagmus of each individual in the head hanging and in the sitting position. From the calculated axes of the slow phase of nystagmus in head hanging and sitting position, they calculated also the angle of these two axes. They proposed angles of more than 130 deg between two axes as representative of a good mirror image. In order to compare the three-dimensional rotation axis of positioning nystagmus to the morphological data, they used the anatomical three coordinates of the semicircular canals measured in the human dry skull as reported by Blanks et al. ( 22). The results of the analysis are as follows. Among the 26 patients tested, 20 patients demonstrated the axes of nystagmus in good or relatively good alignment to the posterior canal axis in the supine position. Nine out of 20 patients exhibited good or relatively good alignment of the axes to the posterior canal axis also in the sitting position ( Fig. 11 upper line). However, in 11 of these 20 patients there was poor alignment of the axis of nystagmus in the sitting position to the posterior canal axis ( Fig. 11 lower line). In addition, six patients showed axes of nystagmus with poor alignment to the posterior canal in the head hanging position. Among them, two patients exhibited axes of nystagmus in good alignment with the anterior canal, in spite of diagnosis of these patients as PC-BPPV from the history and the positioning nystagmus testing. These results indicate that the only-one third of patients, who are diagnosed as PC-BPPV, can be diagnosed as true PC-BPPV. Besides the possibility that the pathology may originate from the anterior canal, it is still unclear which part of the inner ear may be the actual site of the pathology of PC-BPPV in the other 15 patients.
Nystagmus in patients with superior semicircular canal dehiscence
Superior semicircular canal dehiscence (SSCD) syndrome is a newly found entity of vertiginous disease. Minor et al. ( 54) first reported three cases of SSCD. The symptoms of this disease are vertigo and oscillopsia induced by loud sounds (Tullio phenomenon) and by pressure changes, either at the external ear canal or associated with Valsalva maneuver due to a bony dehiscence of the superior semicircular canal at the roof of the temporal bone. Almost all the cases reported showed clear nystagmus during loud sound stimuli and/or Valsalva maneuver ( 54- 56). Yagi et al. ( 57) reported a patient exhibited clear nystagmus evoked by cough, but who did not show any nystagmus by sound stimuli and/or pressure to the external ear canal or Valsalva maneuver. Stimulation creates an inward pressure at the round or oval window results in an outward bulging of the membranous labyrinth in the area of bone dehiscence, with consequent ampulofugal endolymph flow of the superior semicircular canal. On the contrary, stimulation that creates an outward pressure at the round or oval window results in an inward bulging of the membranous labyrinth at the dehiscence with an ampulopetal endolymph flow. Cough increases the intracranial pressure and causes the inward bulging of the membranous labyrinth in the bone dehiscence area of superior semicircular canal. The ampulofugal and ampulopetal endolymph flow produces the excitatory and inhibitory activities of the ampullary nerves, respectively. Therefore, in their case ( 57), the ampulofugal endolymph flow in the right superior semicircular canal occured by coughing and led to an inhibitory nystagmus, which is the downward-clockwise (from subject's view) nystagmus. Minor et al. ( 55) analyzed sound evoked nystagmus three-dimensionally using video-oculography. They found good alignment of sound evoked nystagmus with the axis of superior semicircular canal. Yagi et al. ( 57) also found a good alignment of cough-induced nystagmus with the anatomical axis of superior semicircular canal Fig. 12. From these studies, the pathological localization of the SSCD proposed by Minor ( 54) is proved to be in a superior semicircular canal.
CONCLUSION AND FUTURE ASPECT
From the vector analysis of the slow phase of nystagmus, one can diagnose the localization of the disease in the peripheral labyrinth. However, if the pathology originates from two or three semicircular canals, one hardly can diagnose the detailed localization in the labyrinth, except in very limited cases. In those cases, the strength of the disease should be the same in two or three canals. Then the axis of the nystagmus is the added vector of two or three semicircular canals. Otherwise, the possibility of the axis of nystagmus is located anywhere in the 3D space. Also one doesn't know about the nystagmus related pathological nature of otolith.
We have now quite well developed devices for recording human eye movements. That is video-oculographic devices, which are quite close to the ideal. These devices, however, will be improved upon to more ideal ones in the near future along with the technological advancement. In addition, the engines for analyzing three-dimensional eye movements will be further developed. In these engines, the accuracy of the measurement and the speed of analysis will be improved. Also, these devices with analytical engine will be more cost beneficial for clinical use. Then we, clinicians, can record and analyze the eye movements in patients with vertigo and/or disequilibrium easily.
On the other hand, the anatomical and functional studies, especially about the otolith organs should be carried out. We do have quite limited knowledge about morphology evidences and physiological facts of the otolith organs. The anatomical studies of otolith organs have been done by many investigators. However, a little is known about the detailed three-dimensional structures. Although Uzun-Coruhlu et al. ( 58) demonstrated 3D structures of otolith organs in guinea pigs; still we need more research about the otolith anatomy, especially in humans. From the functional aspect, the ocular counter rolling (OCR), which is generated from the utricle; have been performed since early 20th century ( 59). However, only little basic research regarding otolith-ocular physiology has been performed. Uchino et al. ( 60, 61) observed the monosynaptic and disynaptic connection between the utricular nerve and ipsilateral abducens motoneurons. Also they found polysynaptic circuits from the utricular nerve to inferior oblique and trochlear motoneurons, which cause vertical and rotatory eye movements in the cat. One can interpret these data to understand the otolith-ocular reflex, even though we still have almost no information about the nature of nystagmus, which may come from the otolith organs.
References
1. Baba S, Fukumoto A, Aoyagi M, Koizumi Y, Ikezono T, Yagi T. A comparative study on the observation of spontaneous nystagmus with Frenzel glasses and an infrared CCD camera. J Nippon Med Sch. 2004 2;71(1):25-29. PMID: 15129592.   2. Brandt HF. A bidimensional eye-movement camera. Am J Physiol. 1937 10;49(4):666-670.  3. Wendt Gr. Development of an eye camera for use with motion pictures. Psychol Monograph. 1952;66(7):1-18.  4. Miller EF 2nd, Graybiel A. A comparison of ocular counter-rolling movements between normal persons and deaf subjects with bilateral labyrinthine defects. Ann Otol Rhinol Laryngol. 1963 12;72:885-893. PMID: 14088730.   5. Kumar A, Glenn K. Binocular infrared oculography. Laryngoscope. 1992 4;102(4):367-378. PMID: 1556885.   6. Fender DH. Torsional movements of eye ball. Br J Ophthalmol. 1955 2;39(2):65-72. PMID: 14351667.    7. Shackel B. In: Venables DH, Martin I, editors. Eye movement recording by electro-oculography. Manual of Psycho-physiological Methods. 1967. Amsterdam: North-Holland; p. 299-236.
8. Gonshor A, Malcolm R. Effect of changes in illumination level on electro-oculography (EOG). Aerosp Med. 1971 2;42(2):138-140. PMID: 5547157.  9. Robinson DA. A method of measuring of eye movements using scleral search coil in a magnetic field. IEEE Trans Biomed Eng. 1963 10;10:137-145. PMID: 14121113.  10. Fuchs AF, Robinson DA. A method for measuring horizontal and vertical eye movements in the monkey. J Appl Physiol. 1966 5;21(3):1068-1070. PMID: 4958032.   11. Collewjin H, van der Mark F, Jansen TC. Precise recording of human eye movements. Vision Res. 1975 3;15(3):447-450. PMID: 1136166.   12. Otto D, Gehle F, Eckmiller R. Vide-oculographic measurement of 3-dimensional eye rotation. J Neurosci Methods. 1990 12;35(3):229-234. PMID: 2084392.   13. Yamanobe S, Taira S, Morizono T, Yagi T, Kamio T. Eye movement analysis system using computerized image recognition. Arch Otolaryngol Head Neck Surg. 1990 3;116(3):338-341. PMID: 2306353.   14. Yagi T, Ohyama Y, Kamura E, Shitara A, Kokawa T, Abe S, Nishituji J. New three-dimensional analysis system of eye movements. Otolaryngol Head Neck Surg (Tokyo). 1998;70(4):241-247.
15. Scherer H, Teiwes W, Clarke AH. Measuring three dimensions of eye using dynamic situations by means of videooculography. Acta Otolaryngol. 1991;111(2):182-187. PMID: 2068899.   16. Yagi T, Koizumi Y, Aoyagi M, Kimura M, Sugizaki K. Three dimensional analysis of eye movements using four times high-speed video camera. Auris Nasus Larynx. 2005 6;32(2):107-112. PMID: 15917165.   17. Uemura T, Cohen B. Effects of vestibular nuclei lesions on vetibulo-ocular reflexes and posture in monkeys. Acta Otolaryngol Suppl. 1973;315:1-71. PMID: 4364922.  18. Arai Y, Suzuki JI, Hess BJ, Henn V. Caloric nystagmus in three dimensions under otolith control in rhesus monkeys:A preliminary report. ORL J Otorhinolaryngol Relat Spec. 1990;52(4):218-225. PMID: 2392284.   19. Yagi T, Kurosaki S, Yamanobe S, Morizono T. Three-component analysis of caloric nystagmus in humans. Arch Otolaryngol Head Neck Surg. 1992 10;118(10):1077-1080. PMID: 1389059.   20. Suzuki K. [Three-dimensional analysis of vestibulo-ocular reflexes-the relation between head position and eye movement]. Nippon Jibiinkoka Gakkai Kaiho. 1996 12;99(12):1751-1757. Japanese. PMID: 8997093.   21. Blanks RHI, Curthosy IS, Markham CH. Planar relationships of canals in man. Acta Otolaryngol. 1975;Sep–Oct;80(3-4):185-196. PMID: 1101636.   22. Feter M, Hain T, Zee DS. Influence of eye and head position on the vestibule-ocular reflex. Exp Brain Res. 1986;64(1):208-216. PMID: 3770110.  23. Guedry F Jr. Orientation of the rotation-axis relative to gravity. Acta Otolaryngol. 1965;Jul–Aug;60:30-48. PMID: 14337956.   24. Benson AJ, Bodin MA. Interaction of linear and angular acceleration on vestibular receptors in man. Aerosp Med. 1966 2;37(2):144-154. PMID: 5295433.  25. Hess BJ, Dieringer N. Spatial organization of the maculo-ocular reflex of the rat:Responses during off-vertical axis rotation. Eur J Neurosci. 1990 10;2(11):909-919. PMID: 12106078.   26. Darlot C, Lopez-Barneo J, Tracy D. Asymmetries of vertical vestibular nystagmus in the cat. Exp Brain Res. 1981 2;41(3-4):420-426. PMID: 7215503.   27. Harris LR. Vestibular and optokinetic eye movements evoked in the cat by rotation about a tilted axis. Exp Brain Res. 1987 5;66(3):522-532. PMID: 3609198.   28. Angelaki DA, Hess BJ. Three-dimensional organization of otolith-ocular reflexes in rhesus monkeys. J Neurophysiol. 1996 6;75(6):2405-2424. PMID: 8793753.   29. Cohen B, Helwig D, Raphan T. Baclofen and velocity storage:a method of the effects of the drug on the vestibule-ocular reflex in the rhesus monkey. J Physiol. 1987 12;393:703-725. PMID: 3446808.    30. Darlot C, Denise P, Droulez J, Cohen B, Berthoz A. Eye movements induced by off-vertical axis rotation (OVAR) at small angles of tilt. Exp Brain Res. 1988 10;73(1):91-105. PMID: 3208865.   31. Furman JMR, Schor RH, Schumann TL. Off-vertical axis rotation;a test of the otolith-ocular reflex. Ann Otol Rhinol Laryngol. 1992 8;101(8):643-650. PMID: 1497268.   32. Yagi T, Kamura E, Shitara A. Three dimensional eye movement analysis during off vertical axis rotation in human subjects. Arch Ital Biol. 2000 1;138(1):39-47. PMID: 10604032.  33. Kamura E, Yagi T. Three-dimensional analysis of eye movements during off vertical axis rotation in patients with unilateral labyrinthine loss. Acta Otolaryngol. 2001 1;121(2):225-228. PMID: 11349784.   34. Goldberg JM, Fernandez C. Physiological mechanisms of the nystagmus produced by rotations about an earth horizontal axis. Ann N Y Acad Sci. 1981;374:40-43. PMID: 6978636.   35. Raphan T, Cohen C, Henn V. Effects of gravity on rotatory nystagmus in monkey. Ann N Y Acad Sci. 1981;374:44-45. PMID: 6978641.   36. Raphan T, Schnabolk C. Modeling slow phase velocity generation during off-vertical axis rotation. Ann N Y Acad Sci. 1988;545:29-50. PMID: 3149166.   37. Koizumi Y, Kimura M, Mokuno E, Yagi T. 3D eye movement analysis during sinusoidal off vertical axis rotation in human subjects. Acta Otolaryngol. 2003 1;123(2):121-128. PMID: 12701725.   38. Cohen B, Suzuki JI, Bender MB. Eye movements from semicircular canal nerve stimulation in the cat. Ann Otol Rhinol Laryngol. 1964 3;73:153-169. PMID: 14128701.   39. Suzuki JI, Cohen B, Bender MB. Compensatory eye movements induced by vertical semicircular canal stimulation. Exp Neurol. 1964 2;9:137-160. PMID: 14126123.   40. Della Santina CC, Potyagaylo V, Migliaccio AA, Minor LB, Carey JP. Orientation of human semicircular canals measured by three-dimensional multiplanar CT reconstruction. J Assoc Res Otolaryngol. 2005 9;6(3):191-206. PMID: 16088383.    41. Yagi T, Kamura E, Shitara A. Three-dimensional analysis of pressure nystagmus in labyrinthine fistulae. Acta Otolaryngol. 1999 3;119(2):150-153. PMID: 10320065.   42. McClure JA. Horizontal canal BPV. J Otolaryngol. 1985 2;14(1):30-35. PMID: 4068089.  43. Pagnini P, Nuti D, Vannucchi P. Benign paroxysmal positional vertigo of the horizontal canal. ORL J Otorhinolaryngol Relat Spec. 1989;51(3):161-170. PMID: 2734007.   44. Baloh RW, Jacobson K, Honrubia V. Horizontal semicircular canal variant of benign positional vertigo. Neurology. 1993 12;43(12):2542-2549. PMID: 8255454.   45. Baloh RW, Richman L, Yee RD, Honrubia V. The dynamics of vertical eye movements in normal human subjects. Aviat Space Environ Med. 1983 1;54(1):32-38. PMID: 6830555.  46. Yagi T, Morishita M, Koizumi Y, Kokawa M, Kamura E, Baba S. Is the pathology of horizontal canal benign paroxysmal positional vertigo really localize in the horizontal semicircular canal? Acta Otolaryngol. 2001 12;121(8):930-934. PMID: 11813897.   47. Barany R. Diagnose von Krankhaitserscheinungen im Bereiche des Otolithenapparates. Acta Otolaryngol. 1921;2:434-437.  48. Dix MR, Hallpike CS. The pathology, symptomatology and diagnosis of certain common diseases of vestibular system. Proc R Soc Med. 1952;45:341-354. PMID: 14941845.   49. Schuknecht HF. Cupololithiasis. Arch Otolaryngol. 1969 12;90(6):765-778. PMID: 5353084.   50. Hall SF, Ruby RRF, McClure JA. The mechanics of benign paroxysmal vertigo. J Otolaryngol. 1979 4;8(2):151-158. PMID: 430582.  51. Fetter M, Sievering F. Three-dimensional eye movement analysis of benign paroxysmal positional vertigo and nystagmus. Acta Otolaryngol. 1995 5;115(3):353-357. PMID: 7653253.   52. Aw ST, Todd MJ, Aw GE, McGarvie LA, Halmagi GM. Benign positional nysytagmus. A study of its three-dimensional spatio-temporal characteristics. Neurology. 2005 6 14; 64(11):1897-1905. PMID: 15955941.   53. Yagi T, Koizumi Y, Kimura M, Aoyagi M. Pathological localization of so-called posterior canal BPPV. Auris Nasus Larynx. 2006 12;33(4):391-395. PMID: 16876361.   54. Minor LB, Solomon D, Zinreich JS, Zee D. Sound- and/or pressure-induced vertigo due to bone dehiscence of the superior semicircular canal. Arch Otolaryngol Head Neck Surg. 1998 3;124(3):249-258. PMID: 9525507.   55. Minor LB, Cremer PD, Carey JP, Dera Santina CC, Streubel SO, Weg N. Symptoms and signs in superior canal dehiscence syndrome. Ann N Y Acad Sci. 2001 10;942:259-273. PMID: 11710468.   56. Brantberg K, Bergenius J, Mendel L, Witt H, Tribukait A, Ygge J. Symptoms, findings and treatment in patients with dehiscence of the superior semicircular canal. Acta Otolaryngol. 2001 1;121(1):68-75. PMID: 11270498.   57. Yagi T, Koizumi Y. 3D analysis of cough-induced nystagmus in a patient with superior semicircular canal dehiscence. Submitted.
58. Uzun-Coruhlu H, Curthoys IS, Jones AS. Attachment of the utricular and saccular maculae to the temporal bone. Hear Res. 2007 11;233(1-2):77-85. PMID: 17919861.   59. Barany R. Uber die vom Ohrlabyrinth ausgeloste Gegenrollung der Augen bei Normalhorenden, Ohrenkranken und Taubstummen. Archiv fur Ohrenheilkunde. 1906;68:1-30.  60. Uchino Y, Sasaki M, Sato H, Imagawa H, Suwa H, Isu N. Utriculoocular reflex arc of the cat. J Neurophysiol. 1996 9;76(3):1896-1903. PMID: 8890302.   61. Uchino Y, Sasaki M, Sato H, Imagawa H, Suwa H, Isu N. Utricular input to cat extraocular motoneurons. Acta Otolaryngol Suppl. 1997;528:44-48. PMID: 9288236. 
Fig. 1
Positioning test through infrared CCD camera. The examiner observes the patient's eye movements on a TV monitor. 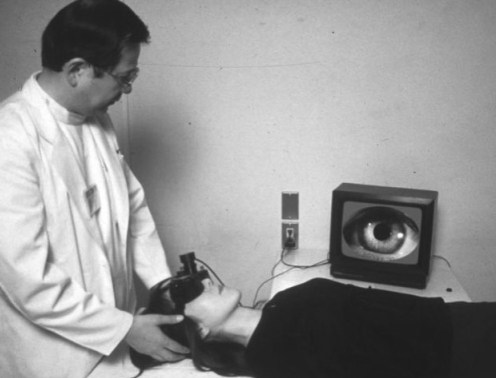
Fig. 2
The simplified schematic representation of scleral search coil method. A uniform alternating magnetic field (vertical arrows) induces an alternating voltage in the eye coil, the amplitude of which is proportional to the sine of the angle between the plane of eye coil and
direction of magnetic field (θ). 
Fig. 3
Infrared CCD camera attached to the specially designed goggle. The goggle for observing the positional and positioning nystagmus through infrared CCD cameras in complete darkness (A) and in light using infrared reflection mirrors (B). 
Fig. 4
Calculation of the horizontal, vertical, and torsional eye movements. The horizontal and vertical eye movements are calculated from the path of the center of the pupil (large +) and the torsional eye movements are calculated from the relation between the several iris striations (small +) and the center of the pupil (large +). 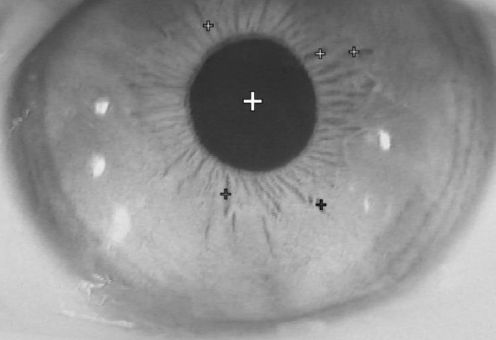
Fig. 5
The printout charts of horizontal (A) and vertical (B) saccades using the high-speed camera of 240 Hz sampling rate. 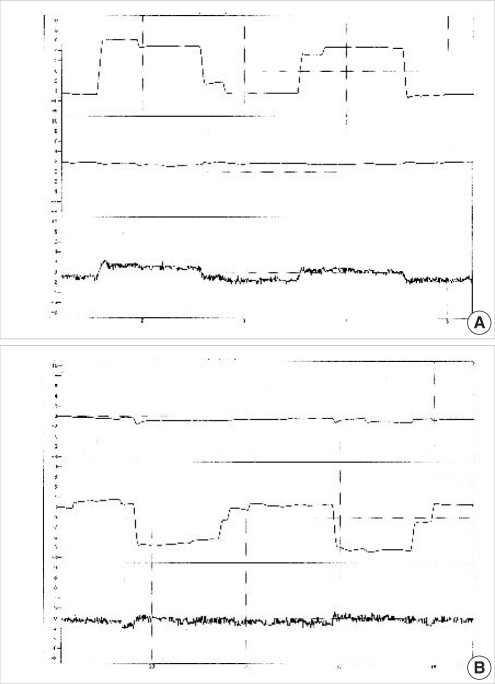
Fig. 6
The average amplitude changes of horizontal, vertical, and torsional eye movements in relation to the head position during a pendular rotation. The filled circles, filled squares, and open diamonds represent the average amplitude of horizontal, vertical, and torsional eye movements, respectively. 
Fig. 7
The hydraulic driven spherical dome for examining the off vertical axis rotation (OVAR). The seen of outside (A) and the inside (B) of the dome. 
Fig. 8
The eye movements during OVAR (A) and traces of the eye position after removing the saccades using computer program (B). 
Fig. 9
The phase of the modulation component (MOC) in relation to the head position (A) and the fitting with least square sinusoids (B). The clear bias component (BIC) is seen in the horizontal eye movement. 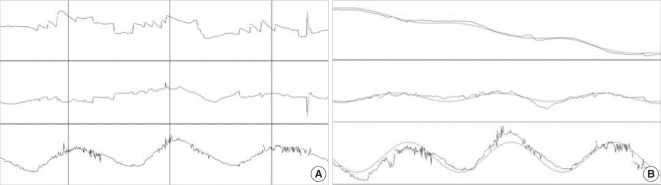
Fig. 10
The three dimensional representation of the velocity vectors of the pressure nystagmus which are recorded from the eleven patients with lateral semicircular canal fistulae due to the cholesteatoma. The velocity vectors of nystagmus in these patients (thin black lines) are well aligned with the anatomical axis of the horizontal canal (circles on the surface of the sphere) with a standard deviation. 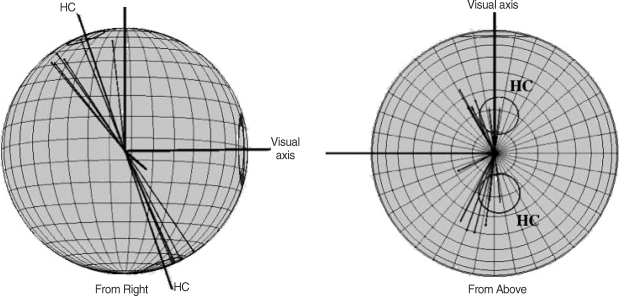
Fig. 11
The axes of the slow phase of nystagmus in a patient showed good alignment to the posterior canal axis both in sitting and head hanging positions (upper line) and that of a patient who exhibited good alignment in head the head hanging position and poor alignment in the sitting position to the posterior canal (lower line). A, B, and C indicate the view of eye from the front, the side, and the above, respectively. LPC: left posterior canal, RPC: right posterior canal. 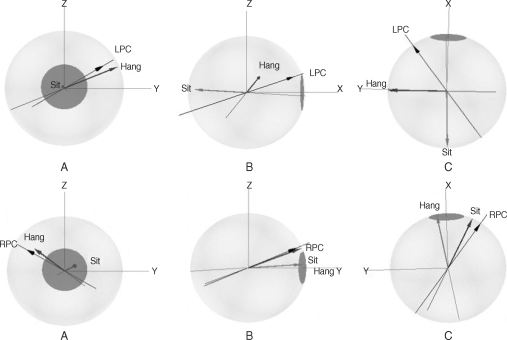
Fig. 12
Three-dimensional representation of the cough-induced nystagmus in a patient with superior semicircular canal ehiscence syndrome. The right side view of the eyeball during cough (A) and same the cough-induced nystagmus as recorded on a different date rom that from A (B). 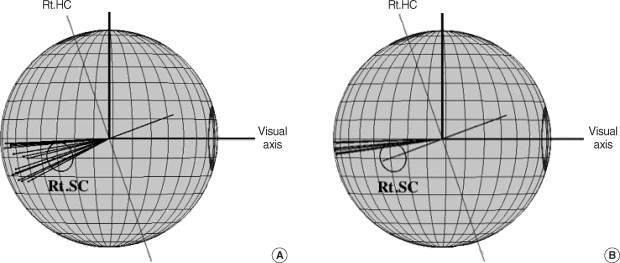
|
|





















