|
 |
- Search
AbstractObjectivesThe aim of this study was to evaluate the clinical features of cochlear reimplantation due to device failure.
MethodsThe medical records of 30 patients who had undergone a revision cochlear implantation were retrospectively reviewed. Causes of revision operations, number of electrode channels inserted, and postoperative speech performances were analyzed.
ResultsDevice failure (N=12, 38.7%) and hematoma (N=3, 9.6%) were the two most common reasons for revision surgery. In patients with device failure, the number of electrode channels reinserted was equal to, or more than the number of channels inserted during initial implantation. Speech performance scores remained the same, or improved after reimplantation in patients with device failure.
ConclusionDevice failure was the most common cause of revision operation in patients with cochlear implanttion. Contrary to expectation, new electrodes were fully inserted without difficulty in all reimplantation cases. Intracochlear damage due to reimplantation appeared to be clinically insignificant.
Cochlear implantation has become the standard treatment for patients with severe to profound sensorineural hearing loss. Moreover, cochlear implantation has been shown to be safe in young children, and is now commonly performed in children younger than 2 years of age (1, 2).
However, revision cochlear implantation are uncommon, and many surgeons are not familiar with revision operations. Nevertheless, revision operation is inevitable in some conditions such as device failure, misplaced electrode, and device migration or extrusion. Wound problems, foreign body reaction, mastoiditis, meningitis, cholesteatoma, and device upgrades may also lead to the need for a revision operation (3-6). Currently, surgeons are being required to conduct more revision operations, because the number of implanted patients is increasing.
During revision surgery, the surgeon should take care not to damage or mobilize the previously implanted device, especially the active electrodes. There are also some restrictions concerning the use of electrocautery. When reimplantation of an active electrode is necessary, the situation becomes more complicated, as removing the initial electrode and inserting a new one may cause intracochlear damage and result in a reduction in speech performance (7, 8). Moreover, the surgeon may be unable to fully reinsert the new electrodes through the previous cochleostomy site (7). Although revision cochlear implantation surgery raises many questions, little is known about its outcomes.
The aims of this study were to evaluate the reasons for reimplantation and the outcomes in patients that had previously undergone cochlear implantation. We placed special emphasis on device failure because this is the most common reason for revision surgery (7-9), and it may carry a higher risk of intracochlear damage following reimplantation.
The medical records of 720 patients that had undergone cochlear implantation between March 1990 and April 2007 were reviewed retrospectively. Patients ages ranged from 12 months to 83 years, and averaged 13.6 years. Of these 720 patients, 30 (4.2%) underwent a revision operation. Two of the 30 had initially received their implants at another institute, while the remaining 28 (3.9%) had undergone initial implantation at the same institute.
There were 12 patients who underwent reimplantation due to device failure. The age of the 12 patients with device failure ranged from 24 months to 30.2 years (average 9.2 years) at reimplantation. Five male and seven female patients were included. All of the patients with device failure who were included in this study had hard failure; there were no soft failure cases. Device failure was confirmed by voltage growth measurements and integrity testing. If a patient presented with no sound perception, the patient was sent to an audiologist for voltage growth measurements by impedance test. If there was no detectable voltage growth, device integrity testing was performed by the manufacturer for confirmation. Device failure was confirmed if there was no response in both the voltage growth measurements and integrity testing.
In reimplantation cases due to device failure, we determined the device type first implanted, with rates of device failure being assessed by the manufacturer. In addition, the number of channels inserted through the cochlea during initial operation was compared to those inserted at reimplantation, in each patient. Speech perception performance was compared before and after reimplantation in cases of device failure, as determined by open-set monosyllabic word and open-set sentence tests. Phonemically balanced words were used for the monosyllabic word test, and Korean version Central Institute for the Deaf (K-CID) score was used for the sentence test. Latest speech perception performance data obtained using initial cochlear implants were compared with latest perception performance data after reimplantation. Changes in speech perception performance were classified as improved, poorer, or no change. An increase in speech perception performance score of greater than 5% was defined as improved. A decrease in speech perception performance score of greater than 5% was defined as poorer, and no change was defined as a change in speech perception performance score of less than 5%.
Statistical analysis was performed using the SPSS software (SPSS Inc., Chicago, IL, USA). The Chi-square test was used to analyze differences between manufacturers with respect to device failure rates. P values of <0.05 were considered statistically significant, and all tests were two-tailed.
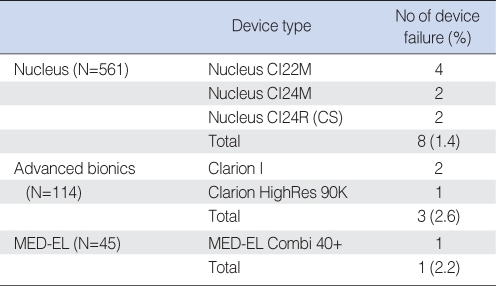
Table 1 presents a summary of the reasons for revision operations. The most common cause of revision operation was device failure (38.7%), and all patients with device failure underwent reimplantation soon after failure had been detected; time lapsed between the two surgeries ranged from 2 months to 9 years 8 months (mean, 38.2 months). The failed devices included 8 (1.4%) Nucleus devices, 3 (2.6%) Clarion devices, and 1 (2.2%) Med-El device (Table 2). No significant difference was found between manufacturers in terms of the incidence of device failure (P=0.42). In device failure patients, we tried to use the most updated model of the same manufacturer's device that was used in the initial implantation. We did not select the device for reimplantation by the size of electrode array. The electrode was incompletely inserted in one patient (Patient 3, 21 electrodes out of 22) at initial implantation. However, electrodes were fully inserted in the other eleven patients (Table 3). At reimplantation, all electrodes were fully inserted in all patients. Table 4 shows result of impedance test after reimplantation in device failure patients.
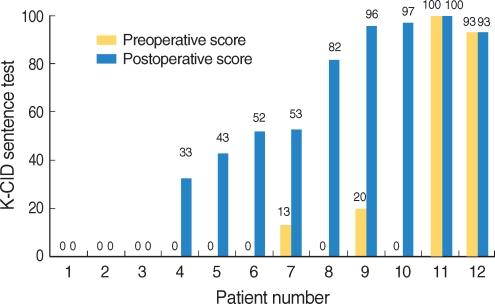
In device failure patients, speech perception performances before and after reimplantation are summarized in Table 5. In the phonemically balanced word test, eight patients (66.6%) demonstrated improved speech perception performance after reimplantation, and four patients (33.3%) demonstrated no change; no patient demonstrated a poorer performance. In the K-CID sentences test, seven patients (58.3%) showed improved speech perception performance after reimplantation, and five patients (41.6%) showed no change. Again, no patient demonstrated poorer scores after revision operation. Fig. 1, 2 demonstrate individual speech perception performance results in patients with device failure, after reimplantation.
The number of patients with cochlear implants that require revision surgery is gradually increasing (9-12). Currently, a revision rate of 5% appears to be a reasonable estimate (9, 13). In the present study, the revision rate was 4.2%, which concurs with previous reports. However, in view of the fact that 3,154 cochlear implantations were performed in South Korea before 2006, it is expected that around 100 of these patients have either undergone or will undergo a revision operation.
Device failure has been reported to occur at an incidence of 3.2-4.9% (12, 14). A multicenter study involving several European cochlear implant centers reported a device failure rate of 3.79% (15), while the present study showed it to be 1.7%. However, the reasons for this lower failure rate are not apparent. In terms of failure rates among manufacturers, previous studies have reported higher rates for Clarion devices than for Nucleus or Med-El devices (16-18). However, in the present study, failure rates were comparable.
When considering reimplantation, one concern is that insertion of the new electrode to the same depth may not be possible. Surgeons are concerned that the intracochlear tunnel may collapse after removing the initial electrode. In fact, one article reported that insertion depth and channel number are significantly lower for second implantations (18). However, other studies found that insertion depth and active channel number remain unchanged after reimplantation (5, 9, 14). In the present study, it was found that the entire length of all electrodes was able to be reinserted in all patients.
Sometimes we observed new bone formation around the posterior tympanotomy site. In such cases, we performed further drilling to remove new bone formation. There was no difficulty in this endeavor, such as resistance to insertion. We did not need to perform further drilling in the cochleostomy site.
According to histological studies on cochlea changes, a fibrous capsule forms around electrodes after insertion (19) and appears to maintain the lumen of the cochleostomy after electrode removal. However, it is not known how long this lumen remains, and, when removing the receiver stimulator, the electrode should be left in situ as a stylet, to maintain the intracochlear lumen until reimplantation, if immediate reimplantation is not possible.
Although hematoma, wound infections, flap problems, and magnet displacement may lead to the need for revision operations, device failure presents a different problems, as it requires intracochlear electrode manipulation during the revision operation. In the present study, we analyzed the speech perception performances of the patients that had experienced device failure, because these patients are at higher risk of intracochlear damage.
If intracochlear structures are damaged during reimplantation, speech perception performance is unlikely to improve despite the presence of a functioning device. In several animal studies, reimplantation has been definitively associated with injury of the organ of corti, stria vascularis, spiral ligament, or spiral ganglion cells (20, 21), and some human studies have reported poorer speech perception after reimplantation (7, 8). Thus, if significant intracochlear damage and no improvement in speech perception performance is expected, it might be advisable not to perform reimplantation based on considerations of the costs of the device and medical services and the risk of complications.
However, recent studies have found that reimplantation may cause little or no damage to inner cochlear structures (22), and the majority of authors have reported that speech perception performance improves after reimplantation (5, 9, 10, 12, 14, 23). In our series, speech perception performances remained the same or improved after reimplantation, and no patient experienced deterioration. Nevertheless, patients 1, 2, and 3 require further explanation, as speech perception performance was 0 in these patients after reimplantation. Patients 1 and 2 had substantial developmental delays, which may have influenced speech perception outcome, whereas patient 3 was lost to follow-up 6 months postoperatively. Thus, it is not clear whether poor speech perception performance after reimplantation was due to intracochlear damage or some other reason in these patients. However, the eight patients whose speech perception performances improved demonstrated that intracochlear damage might not be clinically significant. In general, an improvement in speech perception performance may be expected, and reimplantation should therefore be recommended when device failure is documented.
It should be noted that electrodes removed from our device failure patients were not all modulus hugging type electrodes. During initial implantation in our patients, a conventional electrode was usually located on the lateral side of the scala tympani, which reduces the possibility of spiral ganglion cell damage during reimplantation. However, currently available electrodes are usually of the modulus hugging type, and because this electrode type wraps the weak central core of the cochlea, removing it may cause significant damage to the cochlea. Further studies on reimplantation treatment results in patients who have undergone an initial implantation with a modulus hugging type electrode, are necessary.
Device failure was found to be the most common cause of revision operation in our patients. Full insertion of all channels can be expected during reimplantation, and speech perception performance should improve or remain stable, indicating that intracochlear damage due to reimplantation is clinically insignificant.
References1. Waltzman SB, Cohen NL. Cochlear implantation in children younger than 2 years old. Am J Otol. 1998 3;19(2):158-162. PMID: 9520051.
2. Lenarz T, Lesinski-Schiedat A, von der Haar-Heise S, Illg A, Bertram B, Battmer RD. Cochlear implantation in children under the age of two: the MHH experience with the CLARION cochlear implant. Medizinische Hochschule Hannover. Ann Otol Rhinol Laryngol Suppl. 1999 4;177(1):44-49. PMID: 10214801.
3. Migirov L, Kronenberg J. Magnet displacement following cochlear implantation. Otol Neurotol. 2005 7;26(4):646-648. PMID: 16015161.
4. Migirov L, Yakirevitch A, Henkin Y, Kaplan-Neeman R, Kronenberg J. Acute otitis media and mastoiditis following cochlear implantation. Int J Pediatr Otorhinolaryngol. 2006 5;70(5):899-903. PMID: 16309750.
5. Balkany TJ, Hodges AV, Gomez-Marin O, Bird PA, Dolan-Ash S, Butts S, et al. Cochlear reimplantation. Laryngoscope. 1999 3;109(3):351-355. PMID: 10089956.
6. Windmill IM, Martinez SA, Nolph MB, Eisenmenger BA. Surgical and nonsurgical complications associated with cochlear prosthesis implantation. Am J Otol. 1990 11;11(6):415-420. PMID: 2285061.
7. Miyamoto RT, Svirsky MA, Myres WA, Kirk KI, Schulte J. Cochlear implant reimplantation. Am J Otol. 1997 11;18(6 Suppl):S60-S61. PMID: 9391599.
8. Henson AM, Slattery WH 3rd, Luxford WM, Mills DM. Cochlear implant performance after reimplantation: a multicenter study. Am J Otol. 1999 1;20(1):56-64. PMID: 9918174.
9. Lassig AA, Zwolan TA, Telian SA. Cochlear implant failures and revision. Otol Neurotol. 2005 7;26(4):624-634. PMID: 16015158.
10. Migirov L, Taitelbaum-Swead R, Hildesheimer M, Kronenberg J. Revision surgeries in cochlear implant patients: a review of 45 cases. Eur Arch Otorhinolaryngol. 2007 1;264(1):3-7. PMID: 17009020.
11. Roland JT Jr, Huang TC, Cohen NL. Revision cochlear implantation. Otolaryngol Clin North Am. 2006 8;39(4):833-839. PMID: 16895788.
12. Alexiades G, Roland JT Jr, Fishman AJ, Shapiro W, Waltzman SB, Cohen NL. Cochlear reimplantation: surgical techniques and functional results. Laryngoscope. 2001 9;111(9):1608-1613. PMID: 11568614.
13. Weise JB, Muller-Deile J, Brademann G, Meyer JE, Ambrosch P, Maune S. Impact to the head increases cochlear implant reimplantation rate in children. Auris Nasus Larynx. 2005 12;32(4):339-343. PMID: 15923099.
14. Fayad JN, Baino T, Parisier SC. Revision cochlear implant surgery: causes and outcome. Otolaryngol Head Neck Surg. 2004 10;131(4):429-432. PMID: 15467612.
15. Battmer RD, O'Donoghue GM, Lenarz T. A multicenter study of device failure in European cochlear implant centers. Ear Hear. 2007 4;28(2 Suppl):95S-99S. PMID: 17496657.
16. Hoffman RA, Cohen NL. Complications of cochlear implant surgery. Ann Otol Rhinol Laryngol Suppl. 1995 9;166:420-422. PMID: 7668733.
17. Luetje CM, Jackson K. Cochlear implants in children: what constitutes a complication? Otolaryngol Head Neck Surg. 1997 9;117(3 Pt 1):243-247. PMID: 9334772.
18. Parisier SC, Chute PM, Popp AL. Cochlear implant mechanical failures. Am J Otol. 1996 9;17(5):730-734. PMID: 8892569.
19. Fayad JN, Eisenberg LS, Gillinger M, Winter M, Martinez AS, Luxford WM. Clinical performance of children following revision surgery for a cochlear implant. Otolaryngol Head Neck Surg. 2006 3;134(3):379-384. PMID: 16500431.
20. Simmons FB, Schuknecht HF, Smith L. Histopathology of an ear after 5 years of electrical stimulation. Ann Otol Rhinol Laryngol. 1986;MarŌĆōApr;95(2 Pt 1):132-136. PMID: 3754409.
21. Shepherd RK, Clark GM, Xu SA, Pyman BC. Cochlear pathology following reimplantation of a multichannel scala tympani electrode array in the macaque. Am J Otol. 1995 3;16(2):186-199. PMID: 8572119.
22. Fayad J, Linthicum FH Jr, Otto SR, Galey FR, House WF. Cochlear implants: histopathologic findings related to performance in 16 human temporal bones. Ann Otol Rhinol Laryngol. 1991 10;100(10):807-811. PMID: 1952646.
23. Parisier SC, Chute PM, Popp AL, Suh GD. Outcome analysis of cochlear implant reimplantation in children. Laryngoscope. 2001 1;111(1):26-32. PMID: 11192895.
Fig.┬Ā1Speech perception performance of patients by the Phoneme balanced word test: eight patients (66.6%) demonstrated improved speech perception performance after reimplantation, and 4 patients (33.3%) demonstrated little or no change. 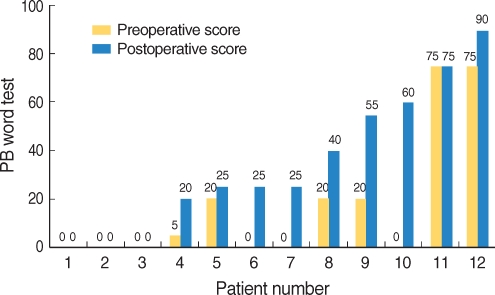
Fig.┬Ā2Speech perception performance of patients by the KCID-sentences test: seven patients (58.3%) showed improved speech perception performance after reimplantation, and 5 (41.6%) showed little or no change.
K-CID test: Korean version of the central institute for the deaf test.
Table┬Ā3Number of channels inserted during first and second implantations in patients with device failure 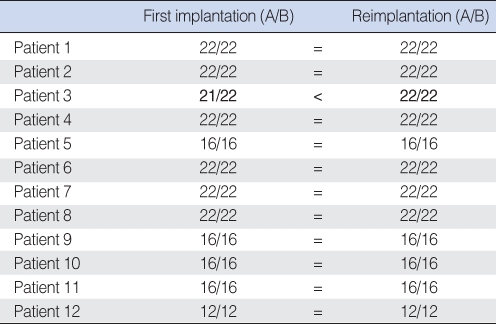
|
|
||||||||||||||||||||||||||||||||||||||||