Sequence Variations and Haplotypes of the GJB2 Gene Revealed by Resequencing of 192 Chromosomes from the General Population in Korea
Article information
Abstract
Objectives
Hearing impairment (HI) is the most common sensory deficit in human. The Gap Junction Protein, Beta-2 (GJB2) gene encodes the protein connexin 26, and this gene accounts for up to half of the cases of autosomal recessive nonsyndromic HI. This study was conducted to obtain a set of sequence variations (SVs) of the GJB2 gene among Koreans from the general population for making molecular genetic diagnoses and performing genetic counseling.
Methods
We resequenced the GJB2 gene in 192 chromosomes from 96 adult individuals of Korean descent and who were without a history of hearing difficulty. The data of the SVs was obtained and the haplotypes were reconstructed from the data.
Results
Five SVs were observed, including a novel one (c.558G>A; p.T186T), with the allele frequencies ranging from 0.5% (1/192) to 41% (79/192). The linkage disequilibrium study and haplotype construction showed that some of the SVs are in tight linkage, resulting in a limited number of haplotypes.
Conclusion
We observed SVs of the GJB2 gene with different allele frequencies, and a limited number of haplotypes were constructed. The data from this study can be used as reference data for GJB2-related hearing genetic studies, including studies on the founder effect and population genetics, and this data is particularly relevant to people of East Asian decent.
INTRODUCTION
Hearing impairment (HI) is the most common form of sensory deficit worldwide, and it affects 0.05% to 0.1% of newborns (1). More than half of prelingual deafness is genetic and it most often shows an autosomal recessive inheritance pattern. Among the many causative genes and loci, the Gap Junction Protein, Beta-2 (GJB2) gene, which encodes the protein connexin 26, accounts for up to half of the cases of autosomal recessive nonsyndromic HI (DFNB1, MIM# 220290) (2). The GJB2 gene is located on the long arm of chromosome 13, band 13q11-12, and it consists of two exons, which are a single protein-coding exon preceded by a noncoding exon. When searching for a gene disease-causing mutation in the GJB2 gene, one may find not only mutations, but also other polymorphisms. Sequence variations (SVs) include mutations, polymorphisms and variations of undetermined significance. A mutation means a disease-causing change and a polymorphism is a non-disease-causing change or change found at a frequency of 1% or higher in the population. A variation of undetermined significance is a variant of unknown clinical significance. A single nucleotide polymorphism (SNP) is a DNA sequence variation that occurs when a single nucleiotide in the genome differs at an appreciable frequency in the population. More than 100 mutations of different types have been reported (http://www.hgmd.cf.ac.uk/ac/gene.php?gene=GJB2) and the spectrum of mutations varies greatly among the different populations with population-specific founder mutations (2-7). While nonsense or frameshift mutations leading to premature truncation are apparently mutational in nature, the disease association of those SVs that leads to single amino acid substitutions (missense or nonsynonymous) has sometimes been confusing and controversial (8, 9), and mislabeling of variants of a nonpathogenic nature would potentially lead to providing incorrect genetic diagnoses and family counseling, as well as interfering with the genotype-phenotype correlations. The segregation patterns of the genotype and phenotype within families and in in vitro studies have been used to support or refute a disease association. In addition, the observation of the homozygous status of a nonsynonymous SV in individuals without apparent HI supports the polymorphic nature of the SV of interest with a minimal phenotypic contribution, although the contribution is not nil. Indeed, several nonsynonymous SVs of the GJB2 gene have been deemed SNPs (http://www.ncbi.nlm.nih.gov/SNP/), while some of them were reported to be mutations (10). Not only are the mutation spectrums different between European descendants and Asian populations, but also the common SNPs and their frequencies are strikingly different between the two ethnic groups according to the genotype data from four populations and that is available at the International Haplotype Mapping Project (Hap Map project; http://www.hapmap.org/). Haplotype is the genetic constitution of individuals with respect to one member of a pair of allelic genes or sets of genes that are closely linked and tend to be inherited together (called linkage disequilibrium [LD]).
By resequencing the GJB2 gene in 96 adult Korean control individuals (192 control chromosomes), we investigated the SVs occurring in the GJB2 gene and we constructed the haplotypes thereof for future studies on the founder effect in the Korean population, and potentially in other East Asian populations as well.
MATERIALS AND METHODS
Resequencing of 96 control Korean individuals
Peripheral blood samples were collected from 96 Korean control individuals (mean age, 53.2 yr; range, 42 to 71 yr) who were recruited from the Health Promotion Center for a routine checkup at our institution. According to the medical records that revealed the findings from a comprehensive evaluation of the health status, none of these study subjects had significant HI. The number of study subjects (96) was based on a previous study that demonstrated that the sample size (number of chromosomes) of 210 would be needed to detect a rare SV occurring at a 1% frequency with 80% power at a 5% significance level (11). Genomic DNA was isolated from the leukocytes using the Wizard genomic DNA purification kit according to the manufacturer's instructions (Promega, Madison, WI, USA). The entire coding exon and the flanking intronic sequences of the GJB2 gene were amplified by polymerase chain reaction using the three pairs of primers designed by the authors (available upon request). Cycle sequencing was performed with the BigDye Terminator Cycle Sequencing Ready Reaction kit (Applied Biosystems, Foster City, CA, USA) on the ABI 3100 Genetic Analyzer (Applied Biosystems). Sequence variations were analyzed with reference to the wild type sequence using the Sequencher program (Gene Codes Corp., Ann Arbor, MI, USA). The identified variations were described according to the guidelines by the Human Genome Variation Society (http://www.hgvs.org/mutnomen/).
Pairwise linkage disequilibrium and haplotype analyses
Pairwise linkage disequilibrium (LD) between the SVs with minor allele frequencies (MAF) greater than 2% was analyzed by two parameters, |D'| and r2. D is a commonly used coefficient for LD. D' is introduced to normalize D by dividing it with the theoretical maximum for the observed allele frequencies (D'=D/Dmax). r2 is another common way of representing LD (12). We used the graphical overview of linkage disequilibrium (GOLD) statistical package to obtain |D'| and r2 (13). The haplotypes from the SVs were reconstructed by the PHASE program, ver. 2.1.1. (14).
RESULTS
The percent coverage of the resequencing of the GJB2 gene was 100% in the 192 control chromosomes. We found a total of five SVs; four nonsynonymous SVs and one novel synonymous SV (Table 1). The two most commonly occurring nonsynonymous SVs were V27I and E114G. V27I was observed in the heterozygous state in 49 individuals and in the homozygous state in 15 with a MAF at 0.411. E114G was observed in the heterozygous state in 27 individuals and in the homozygous sate in 5 with a MAF at 0.193. Two individuals were shown to be heterozygous for V37I (MAF, 0.010), and four individuals were heterozygous for T123N (MAF, 0.021). Five individuals were heterozygous for I203T (MAF, 0.026). We found a novel synonymous SV, T186T, from 558G>A in a single individual. The genotype frequencies of each of the above SVs were in Hardy-Weinberg equilibrium.

Sequence variations of the GJB2 gene and their observed frequencies in 192 chromosomes from the general population in Korea as compared with the data from previous studies
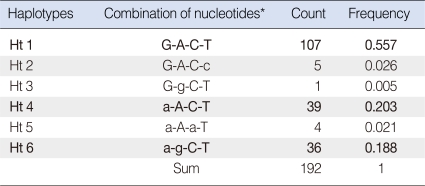
The analysis of pairwise LD between the SVs with a MAF greater than 0.01 (1%) showed that the two most frequent SNPs, V27I and E114G, are tightly linked (|D'|=0.943, r2=0.304) (Table 2). In addition, T123N and I203T are in complete linkage with V27I. This LD profile is reflected by the results from the haplotype analyses (Table 3). The inference of haplotypes by the PHASE algorithm reconstructed six haplotypes (Ht 1-Ht 6) from the genotype data of the four loci with MAFs greater than 1% (V27I-E114G-T123N-I203T in order by the physical location). The common haplotypes occurring at frequencies greater than 10% (0.1) in the 96 control Koreans were Ht 1 (G-A-C-T; frequency, 0.557), Ht 4 (a-A-C-T; frequency, 0.203) and Ht 6 (a-g-C-T; frequency, 0.188) (bold; major alleles are in capitals). This shows that the E114G variant was occurring in tight linkage with V27I, which was also supported by the high LD values between the two loci. The other rare haplotypes also show that T123N was occurring exclusively in alleles with V27I (a)-E114E (A)-I203I (T) (Ht 5), and I203T was occurring in alleles with V27V (G)-E114E (A)-T123T (C) (Ht 2).
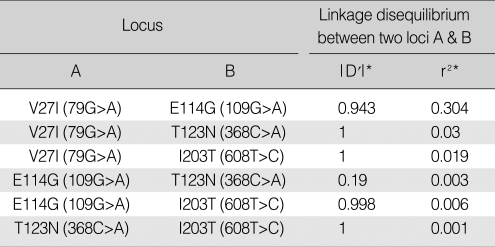
Pairwise linkage disequilibirum between the four loci with minor allele frequencies greater than 0.01 (1%)
DISCUSSION
More than 50% of prelingual deafness has a genetic background that is most often autosomal recessive and nonsyndromic. Despite the extreme genetic heterogeneity, the GJB2 gene has been reported to account for roughly half of autosomal recessive nonsyndromic HI in European descendants and there is a high rate of mutation carriers in the general population due to a founder mutation, 35delG (6). GJB2-related hereditary HI also occurs in other populations, with 235delC being a prevalent mutation in East Asians, including Koreans (7, 15-17).
The variant with the highest frequency in our data was V27I, with 15 individuals having the homozygous genotype. V27I is a common variant that exclusively occurs in Asian populations (Table 1) (4, 18). Previous reports have also described V27I in the homozygous state in control individuals, and it has the reference SNP number rs2274084 (16). Another common variant, E114G, has also been assigned the reference SNP number rs2274083. Although its disease association has been controversial, the occurrence of homozygous E114G in a sufficient number of control individuals in this study, as well as in other studies, strongly supports its polymorphic nature (4, 16, 18). According to the HapMap project and the dbSNP database at the NCBI website (http://www.ncbi.nlm.nih.gov/projects/SNP/), V27I and E114G are haplotype-tagging SNPs. The HapMap data shows that these two SNPs are observed only in Han Chinese and Japanese, and not in European descendents or Africans. We also noted that the MAF of E114G in Koreans from our data (0.193) falls in between those of Chinese (0.289) and Japanese (0.102), while the MAF of V27I in Koreans (0.411) is comparable to those in Chinese (0.375) and Japanese (0.398). However, a recent multi-center dataset still labeled V27I and E114G as mutations (10). Three rare nonsynonyous variants (V37I, T123N and I203T) were observed in this study only in the heterozygous state. These variants have also been rarely reported in other populations (4, 18-20), and they have been designated as missense mutations according to the mutation database. We found a novel synonymous SV in codon 186 in a single individual (heterozygous for T186T). Whether T186T is an extremely rare, silent SNP or it is a deleterious mutation resulting from aberrant splicing is unknown. There were no alleles with deleterious mutations, including 235delC, which is the most common mutation in East Asian populations, including Koreans, and in our control individuals (4, 16).
Since the phenotypic spectrum of GJB2 mutations encompasses early-onset, prelingual deafness to late-onset deafness, the control individuals in our study, all of whom were adults (age>41 yr), are thought to better represent a control population for the GJB2 gene than the control population in the previous study, which involved newborns with normal audiologic screening as control subjects (16). However, it was a limitation of this study that we could not provide objective audiologic data for the study subjects; we labeled them as controls based on their medical records that showed no evidence of HI being documented according to their comprehensive health status evaluations. Last, the data from this study showing tight linkage between SVs and the limited number of haplotypes occurring in the Korean population can be used as reference data for studies on GJB2-related hearing genetics, including the founder effect and population genetics, and the data is particularly relevant to people of East Asian decent.
Notes
No potential conflict of interest relevant to this article was reported.
ACKNOWLEDGMENTS
This study was supported by the Samsung Medical Center Clinical Research Development Program grant, #CRS-106-56-2.