Improving the Accuracy of Baha® Fittings through Measures of Direct Bone Conduction
Article information
Abstract
Objectives
Variability in Baha® sound processor fittings may arise from the nature of the implant-to-bone transmission as well as transcranial attenuation for patients with single-sided sensorineural deafness (SSD). One method of improving the predictability of Baha fittings is to measure the individual patient's actual bone conduction thresholds, thereby removing the influences of skin thickness and/or the implant location site.
Methods
Twenty adult wearers of the Baha bone conduction implant system participated in the study. Direct bone conduction thresholds were obtained through the BC Direct function of the Baha Fitting Software combined with the Cochlear Baha BP100 sound processor. For comparison, the masked and unmasked bone conduction responses of the patients were collected through standard audiometric testing techniques. Test-retest reliability measurement was performed for all participants. Data for each frequency and frequency range were analyzed separately.
Results
The results confirm the improved transmission of sound through the implant rather than transcutaneously through the skin. On average, the BC Direct thresholds were closer to the patient's unmasked thresholds than the masked values. In subjects with SSD, BC Direct results were poorer than contra-lateral bone conduction thresholds, most likely due to transcranial attenuation. The test-retest reliability for the BC Direct measurements was within +/-5 dB. The comparison of preferred amplification, based on direct bone conduction or bone conduction audiometry, found higher agreement for fittings based on direct bone conduction measurements.
Conclusion
While the transfer function between the implant and the skin can be predicted on average, there are a number of patients for whom measurement is essential to determine the required amplification. These were patients with: 1) SSD, 2) asymmetrical hearing loss, 3) unusual implant location or skull formation, and 4) users of Testband or Softband. The result for the clinician is that a fitting can take place with less fine-tuning and a greater understanding of the variability of bone conducted sound transmission.
INTRODUCTION
Modern osseointegrated bone conduction hearing systems, such as Baha®, are fitted based on the patient's audiometric hearing thresholds. However, an important limitation when measuring a patient's bone conduction thresholds using an audiometer is that the transmission of vibrations must pass through the skin to the cochlea in the temporal bone. Furthermore, in the case of single-sided sensorineural deafness (SSD), the sound must continue through the temporal bone to the contralateral cochlea with approximately 5-10 dB further interaural attenuation (1-3). Of concern is that the variability between or within patients is between 0-30 dB at any given frequency (1). This attenuation of sound through the skin and bone adds to the variability in a Baha fitting and if not accounted for may lead to the patient being under- or over-amplified. Potentially, this could result in poorer than predicted performance with the Baha sound processor (2, 4-7). Therefore, if we wish to increase the accuracy of the initial fitting, clinicians require a mechanism to easily and directly measure the bone conduction thresholds in an individual patient.
A proposed solution is to generate test signals from the Baha sound processor and transmit these directly to the cochlea via an osseointegrated implant. This test condition has been labeled direct bone conduction (DBC) (8).
In terms of developing a clinical application, Stenfelt and Hakansson (4) proposed three measures that could be used to measure the direct bone conduction in a patient. The first is to measure the force or acceleration required. While this is advantageous because it is independent of the transducer, this type of measurement is difficult to perform clinically. The second is to measure the patient's free-field hearing thresholds. This would involve measurement of the entire Baha system, thereby increasing the variability, and it could be confounded by the impact of non-linear signal processing such as expansion and compression. Third is to directly measure the voltage driving the transducer. In this case, the desired voltage can be converted to force thresholds and a corresponding levels in dB HL.
Similar to the third proposal, the authors developed a procedure applicable to the clinical setting. Here, an inbuilt digital signal generator in the Baha device can be used to measure the patient's thresholds. The digital values required to produce the threshold can then be recorded by a connection to a computer using a standard interface used by hearing aids, i.e., Hi-Pro, Noah Link, or similar (9). The key advantage of this method is that all tolerance chains normally experienced between measurement equipment and device setups are eliminated.
The aim of the present investigation is two-fold. The first is to validate, with a new Baha sound processor, the previously published reference equivalent threshold force level for DBC (10). The second is to determine the clinical need for measuring DBC values through a Baha sound processor.
MATERIALS AND METHODS
Subjects
Twenty adults with a skin penetrating titanium implant for standard attachment of a Baha participated in this study (Table 1). The subjects were patients with conductive or mixed hearing loss. Each subject was selected for the Baha procedure in line with internationally accepted patient selection criteria (11). The study was conducted following good clinical practice (GCP) with approval from the Regional Ethical Review Board in Gothenburg.
Materials
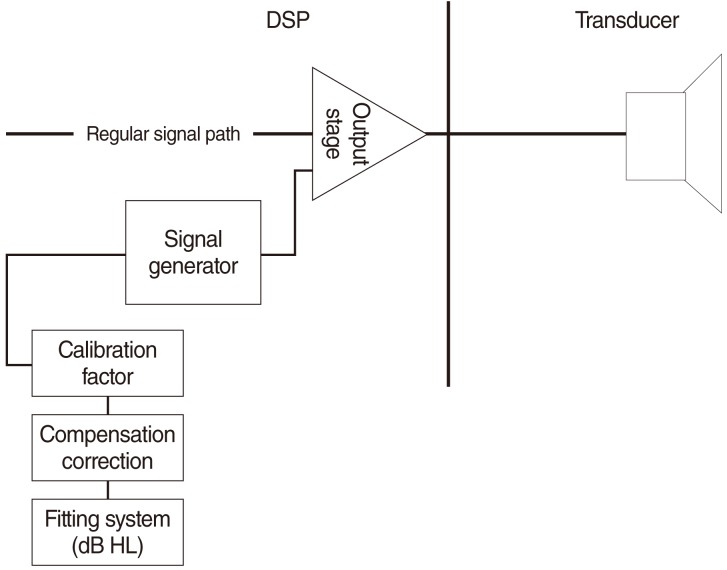
The test device used was the Cochlear Baha BP100 (Cochlear Bone Anchored Solutions AB, Gothenburg, Sweden) sound processor in combination with the Cochlear Baha Fitting Software ver. 1.0. The combination allows the use of a software feature called BC Direct to measure direct bone conduction. The patient's responses to the vibratory signals by the Baha sound processor are obtained (Fig. 1). The Fitting Software allows the frequency (Hz) and level (dB HL) of those sounds to be controlled.
The device signal generator and connected computer system were calibrated so that, for a given frequency, a dB HL value read in the Cochlear Baha Fitting Software ver. 1.0 would correspond to the device force level outputs suggested by Carlsson et al. (10). As audiologists are most familiar with the dB HL method of measurement, the scale provided is in dB HL.
The maximum output force value of the device is measured on a skull simulator. The force level needed to convert to dB HL is then subtracted from the maximum output. This gives the maximum output value in dBHL that the device can produce.
If a lower level of stimuli is desired, the digital control values in the device are reduced by an equal amount. This can be done since a subtraction in the logarithmic domain is the same as a division in the linear domain, and hence, the variable control value in the device will have no unity. The resulting control value will be a "relationship" between the maximum output and the desired output.
Device control value=-(Maximum output value-Desired stimuli)
Equation 1. Calculation of device control value
The values in dB HL are then converted to digital values in the sound processor and produced by the device in dB rel. 1 µN (Equation 1). The pure-tone is generated at a set frequency at each octave and half-octave between 250-8,000 Hz. The complete system has been verified by the manufacturer (Cochlear Bone Anchored Solutions AB, Cochlear Ltd, Gothenburg, Sweden) to meet the audiometer standard (ISO 60645-2) with regard to allowable tolerances in terms of level (dB) and frequency (Hz). The audiologist can use standard audiological threshold seeking techniques to measure the softest response for a given frequency and select these values to be used as the basis for the hearing loss rather than the standard audiometric test rests.
Procedure
Audiometric evaluation consisting of air and bone conduction testing was performed in accordance with ANSI S3.21 in a sound insulated test room using a Madsen Conera Audiometer (GN Otometrics, Copenhagen, Denmark). For comparison purposes, bone conduction thresholds were obtained at 250, 500, 1,000, 2,000, 3,000, 4,000, and 6,000 Hz with narrow-band noise applied via air conduction to the contralateral ear when necessary. For the measurement of DBC, the Cochlear Baha BP100 sound processor was used in conjunction with the Cochlear Baha Fitting Software ver. 1.0. Measurement was conducted using the same audiometric techniques as specified in ANSI S3.21 with the Baha sound processor microphone turned off. This determines the softest sound at which the patient can respond through DBC. It should not be confused with the measurement of an actual ear specific threshold, as masking is not used in this measurement.
Determining reference equivalent force threshold
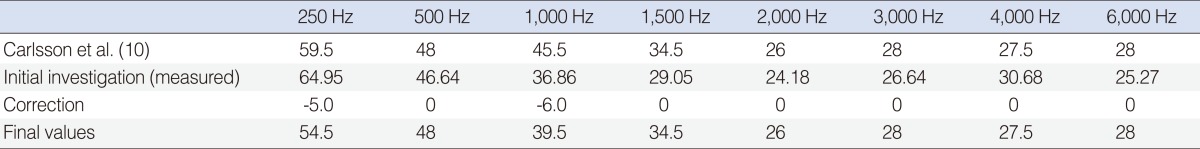
For the reference equivalent force threshold, the starting point was the values recommended by Carlsson et al. (10). With the BP100 sound processor, it is possible to measure DBC at 6,000 Hz and provide amplification at 6,000 Hz. Unfortunately, the previous study did not measure 6,000 Hz. Based on ISO-389-3:1994, which shows an increase of 4.5 dB in reference to an equivalent threshold force level at 6,000 Hz over 4,000 Hz to compensate for the additional skin impedance (13) (i.e., no net effect for direct bone conduction), we decided to keep 6,000 Hz equivalent to 4,000 Hz. For the pilot study, 11 adult Baha patients participated. We compared the measured unmasked bone conduction measurements with the DBC measurements. For all frequencies except 250 Hz and 1,000 Hz, we found that the BC and DBC thresholds were within +/-5 dB, which is the acceptable error rate. In line with a study by van der Pouw et al. (14), we decided to correct the values at 250 Hz by 5 dB and at 1,000 Hz by 6 dB. The extrapolated value at 6,000 Hz showed good correspondence with the measured value (Table 2).
RESULTS
Examination of the results led to a number of interesting comparisons. First, we compared the measured BC Direct results with the measured masked bone conduction responses (Fig. 2). Masked bone conduction responses were selected for comparison, as these are what the audiologist would have readily available from the audiometric testing and what hearing aid databases, such as Noah, use as the default bone conduction values. Therefore, this analysis allows us to compare DBC values with standard clinical practice, as recommended by American Speech-Language-Hearing Association (ASHA) (15).
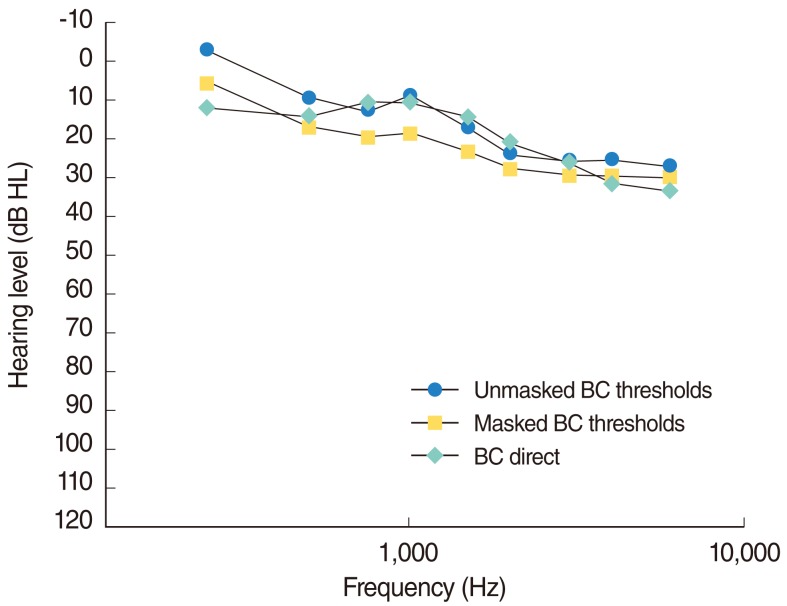
Comparison of the measured masked and unmasked bone conduction thresholds versus measured BC Direct responses across a group of 20 participants.
Fig. 2 demonstrates that, on average, the measured values were consistently 5-10 dB better across the frequency range than the patient's bone conduction thresholds in the test ear. This variation is outside of the normally accepted audiometric error rate of +/-5dB.
One reason for the difference between measured BC Direct and bone conduction thresholds may be due to measurement error. To rule this out, we investigated the repeatability of the BC Direct procedures and found that across the group, the repeatability (across two separate visits) for BC Direct was within +/-5dB of the previously measured values. Therefore, BC Direct provides a repeatable method of measuring the softest sound produced through the Baha sound processor.
To further understand the performance, we investigated why, on average, the BC Direct values might be better than those predicted from the pure-tone audiogram. In examining the data, there were four groups of participants for whom additional investigation was required. The first group was comprised of patients with asymmetrical hearing loss, where the Baha is placed on the side with worse sensorineural hearing thresholds. In this case, if the patient has sufficiently low interaural attenuation, then the non-test cochlea might hear the sound first. Second, for some patients, both cochleae may hear the sound at the same time, thus allowing loudness summation. Third, for single-sided sensorineural deafness, the high variability in interaural attenuation might cause difficulties with prediction. Fourth, due to implant placement or other anatomical variables, the predicted values may differ from actual values (7).
Looking at the possible reasons for this difference, one situation requires further investigation. A number of the possibilities include the scenario where the side with the Baha implant may have poorer hearing thresholds than the non-Baha side. In this situation, even with transcranial attenuation, it is possible that the contralateral cochlea could be responding when tested in a sound booth. Therefore, the assumption made in a previous study (10) that masked and unmasked thresholds can be treated equally deserves further investigation. In order to investigate this, we compared the masked and unmasked bone conduction results with the DBC values (Fig. 2).
The relationship between DBC values and unmasked bone conduction results was higher than for masked bone conduction results. The DBC and unmasked bone conduction results were within 5 dB at all frequencies, except for 250 Hz and 6,000 Hz. The differences at 250 Hz are most likely due to vibro-tactile difficulties in measuring bone conduction. The differences at 6,000 Hz are most likely due to calibration issues for transmission of high frequency tones via a standard bone conductor.
DISCUSSION
For most patients, the increase in transmission efficiency between transcutaneous and percutaneous transmission can be accurately estimated using published results (10). For some patients, however, a number of factors (e.g., significantly better bone conduction thresholds in the non-Baha ear, use of clinical masking, differences in skin thickness or placement of the implant) may result in the measured DBC to be larger or smaller than predicted. Additionally, the data discussed is based on averages, and there is between-subjects variability in this study and previous studies in terms of DBC values.
Previous studies (10) have used patients who had the implant in the best ear and assumed that there was no need for masking and the difference between the BC and DBC thresholds is the same with or without masking. Based on their data, the authors concluded that either the masked or unmasked information could be used. The data from the current study indicates that for some patients, the Baha may be placed in the ear with poorer cochlea thresholds and that some caution should be taken in assuming that unmasked and masked bone conduction thresholds are the same. Therefore, to ensure accuracy, the unmasked bone conduction thresholds should be used.
Based on the current study, one could make the argument that simply taking the unmasked bone conduction thresholds would make redundant the use of tools such as BC Direct. Unfortunately, it is common that in the patient's chart, only the masked values are recorded, because the unmasked values are considered to be 'noise.' Similarly, while the average values are correlated in a manner similar to other areas in audiology, such as the measurement of real ear to coupler difference in hearing aid fitting, the individual results contain uncontrolled variability.
Direct measurement of bone conduction thresholds is recommended for all Baha fittings and especially those cases involving:
- Patients with asymmetrical bone conduction thresholds where the Baha is placed in the side with the poorer cochlea function.
- Patients with single-sided sensorineural deafness due to the variability in interaural attenuation.
- Patients using the soft-band or head-band due to variability in transcutaneous transmission (16).
- Patients where the Baha implant was placed in a non-optimum position or with significant craniofacial abnormalities.
In each of these scenarios, the difference between the actual and predicted direct bone conduction thresholds may result in the patient being under- or over-amplified and potentially result in poorer than predicted performance with the Baha sound processor. Given the complexity of correcting the audiogram versus the simplicity and speed of measuring BC Direct, BC Direct provides an improved method of compensating for the variability in the individual Baha patient.
From the results obtained in the current study, we can conclude that if a patient has better cochlea function in the non-Baha ear, then this will add into their perception with DBC measurements. However, the clinician may not record unmasked BC thresholds in these patients audiometrically but rather record only document masked thresholds. Therefore, a fitting using the audiometric (masked) thresholds would result in greater gain than needed and greater gain than a fitting done with DBC. Given the complexity of correcting the audiogram versus the simplicity and speed of measuring BC Direct, BC Direct provides an improved method for compensating for the variability in the individual Baha patient.
BC Direct provides a validated method of comparing the transcutaneous thresholds as measured through audiometry with the percutaneous responses from the Baha sound processor. These values from BC Direct can then be used in preference to the patient's bone conduction thresholds for the prescription of amplification.
Notes
The authors are both employees of Cochlear Bone Anchored Solutions AB.