Evaluation of the Interleukin-1 Receptor Antagonist and Immunoregulatory Interleukin-10 in the Middle Ear in Chronic Otitis Media With Effusion in Children With and Without Atopy
Article information
Abstract
Objectives
The role of pro-inflammatory cytokines in the course of chronic otitis media with effusion (COME) has been documented. However, there are fewer studies on the action of anti-inflammatory cytokines in the middle ear. We sought determine whether there is an association between COME and anti-inflammatory cytokines and whether there are any differences in the cytokine profile in COME children with and without atopy.
Methods
Eighty-four children were divided into 3 groups: 32 nonatopic children with COME (group NA), 31 atopic children with COME (group A), and 21 children without COME and without atopy (control group C). Specimens from the middle ear were collected and evaluated by enzyme-linked immunosorbent assay for the cytokines interleukin-1 receptor antagonist (IL-1Ra) and immunoregulatory IL-10.
Results
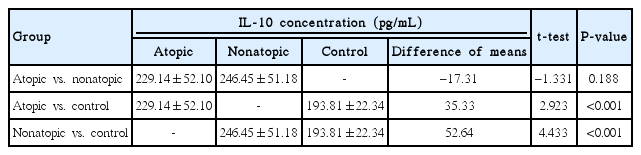
Significantly higher IL-10 concentrations were found in both nonatopic and atopic children with COME compared to controls. No significant differences in IL-1Ra levels were found between atopic and nonatopic children with COME and the control group.
Conclusion
We found no differences in the levels of IL-1Ra in atopic and nonatopic children with COME compared to controls. However, we found elevated IL-10 levels in the middle ear effusions from children with COME, with or without atopy. These elevated immunoregulatory cytokine levels suggest a role for new immunomodulatory treatments to prevent disease progression in COME, regardless of atopy.
INTRODUCTION
Otitis media with effusion (OME), also known as secretory otitis, is a childhood disease that has become increasingly more common in the last decade. This condition may lead to severe irreversible changes within the middle ear, leading to conductive hearing loss with subsequent impairment of speech and language development.
Chronic otitis media with effusion (COME) relates to the permanent presence of secretion in the middle ear for more than 3 months and, compared to acute otitis media (AOM), it is usually asymptomatic. The causes of COME are not fully understood, and current treatment approaches do not resolve the underlying inflammation. There are also discrepancies in individual responses to the available treatments, which are suggested to be due to variations in the course of COME depending on the presence or absence of atopy in the children.
While the exact pathophysiology of OME is unclear, bacteria and viral infections, as well as cellular and humoral immune responses, are known to play a role. In particular, numerous studies have confirmed the local involvement of pro-inflammatory cytokines in the ongoing pathological process in children with OME [1,2]. While disruption of the regulation of the pro-inflammatory cytokines tumor necrosis factor (TNF) α, TNFβ, interleukin (IL)-1β, interferon γ, IL-6, and IL-8 in OME patients has been well documented [3], the role of anti-inflammatory cytokines may be just as important. However, there are few studies evaluating the action of anti-inflammatory cytokines, including IL-10 and IL-1 receptor antagonist (IL-1Ra), in the middle ear of children with OME.
IL-1Ra is a member of the IL-1 cytokine family, and is responsible for maintaining a tight balance between immune response against infection and uncontrolled inflammation (for a recent review see [4]). Children born with a genetic deficiency of IL-1Ra suffer from severe systemic and local inflammation [5], and disrupting the delicate balance between IL-1 and IL-1Ra has been liked with many diseases [6]. IL-1Ra binds to the interleukin-1 receptor, thus preventing IL-1 binding to this receptor, abolishing its biological effects [7] and preventing the spread of inflammation. IL-1Ra also plays a role in allergy; excess IL-1Ra has been observed in the nasal discharge of healthy individuals compared to a group of patients with allergic rhinitis [8]. Similarly, a strong correlation was established between the duration of exposure to the allergen and increased concentrations of IL-1β and IL-1Ra [9]. Because of its role in allergy, the levels of IL-1Ra may differ in atopic and nonatopic patients with OME, and may affect disease progression.
There is also potential for IL-1Ra to be used as a treatment for OME. It has been shown to decrease inflammation in mice [10], and a recent study reported success in using IL-1Ra as a vaccine in patients with rheumatoid arthritis [11]. Other studies have confirmed the role of IL-1Ra as a modulator in inflammatory conditions, highlighting its potential for application in clinical practice [12].
While the levels of IL-1Ra in middle ear effusions during OME is unclear, an increase in IL-10 levels in middle ear effusions has already been reported [11-16]. IL-10 is proposed to act as a potent anti-inflammatory agent by downregulating cytokine expression, inhibiting cytokine release and shortening the duration of immune responses that accompany inflammation (reviewed in [17,18]). Despite this, the exact role of IL-10 in progression of OME and its potential as a therapeutic target remains unclear.
Confirming the involvement of both IL-10 and IL-1Ra in OME would aid our understanding of the inflammatory response underlying this condition, and help us to identify new immunomodulatory drugs to halt disease progression. The aim of the study was to determine whether locally produced anti-inflammatory cytokines are involved in COME and if there are any differences in the cytokine profiles between atopic and nonatopic children.
MATERIALS AND METHODS
Patients
The study was conducted in 84 children, who based on history, physical findings, results of accessory investigations (allergic symptoms, total serum IgE and allergen-specific IgE concentrations, provocation tests), and similar clinical and audiological parameters, were assigned into one of 3 groups. The nonatopic group (group NA) included 32 children (mean age, 6 years 7 months) with COME but without atopy, group A consisted of 31 atopic children (mean age, 6 years 2 months) with COME, while the control group (group C) was comprised of 21 children (mean age, 6 years 2 months).
The diagnosis of OME of more than 3 months’ duration (i.e., COME) was confirmed by characteristic otoscopic and/or microscopic findings, type B tympanometric curve, conductive hearing loss with the cochlear reserve of >30 dB, and no episodes of AOM in the past 3 months. In patients with COME (groups A and NA), ventilation tubes were inserted for treatment.
The diagnosis of atopy (group A) was determined by measuring both the total and specific serum concentrations of class E antibodies (IgE) specific for the following allergens: dust mites (Dermatophagoides pterygonyssinus or Dermatophagoides farina), fungus (Alternaria or Cladosporium), the epidermis of animals (i.e., dog or cat), and grasses, grains, trees and weeds. Total IgE serum concentration was determined by enzyme-linked fluorescent immunoassay or ELFA (an indirect noncompetitive immunoenzymatic sandwich method using a murine monoclonal anti-IgE with fluorescent detection of the final product in IU/mL). Specific IgE in the serum were determined by enzyme-linked immunosorbent assay or ELISA method (an indirect noncompetitive immunoenzymatic sandwich method using the antigen [allergen] coated on the solid phase with colorimetric detection of the final product of the substrate in IU/mL). The standard for the total serum IgE concentration was greater than 150 IU/mL. The presence of specific IgE class antibodies in the serum against the above allergens were described as classes 0 to 5. In all patients diagnosed with atopy, these values were above class 3.
The control (C) group consisted of children without symptoms of OME and with normal total IgE antibody levels in the serum and no specific IgE antibodies against the studied allergens. In these children, diagnostic puncture the eardrum (myringotomy) was performed due to obstruction of the eustachian tube caused by adenoid hypertrophy, as confirmed by a type C tympanogram. However, if there was no exudation in the tympanic cavity during this diagnostic puncture of the tympanic membrane, the patients were included in group C.
None of the children examined were receiving antiallergic drugs, corticosteroids or antibiotics in the last 3 months prior to surgery (i.e., ventilation tubes [groups A and NA] or myringotomy [group C]). However, prior to the last 3-month period, patients in groups A and NA were treated conservatively with antiallergic drugs and/or corticosteroids and/or antibiotics, albeit unsuccessfully. The patients in the control group were not under conservative treatment due to adenoid hypertrophy.
In all children, their history, physical characteristic, and results of accessory investigations, were recorded. The characteristics of the subjects are summarized in Table 1.

Characteristics all children included in the study (atopic and nonatopic children with COME, compared to the control group)
The Bioethics Committee at the Military Institute of Medicine in Warsaw, Poland approved the study.
Methods
The effusions from the tympanic cavity were collected into sterile test tubes (from children with COME) or the tympanic cavity was irrigated with 0.5 mL of sterile saline using a suction set (in controls). The material obtained from the tympanic cavity was frozen at –80°C for further studies of the interleukins IL-1Ra and IL-10.
The ILs in middle ear specimens were studied at the Immunology Laboratory, Microwave Protection Department, Military Institute of Hygiene and Epidemiology in Warsaw. The most active interleukins participating in anti-inflammatory responses were selected for the study. Their concentrations were determined in the supernatant obtained by centrifugation from middle ear specimens, using ELISA. The enzyme activity of the complex was evaluated using an ELX-800 analyzer (Bio-Tek Instruments Inc., Winooski, VT, USA). Optical density was measured at a wavelength of 450 nm. Interleukin activities were determined by measuring their concentrations in the recovered and centrifuged fluid using R&D Systems Inc. (Minneapolis, MN, USA).
IL concentrations were determined by setting up a standard curve, measuring the absorbance, and plotting absorbance versus standard concentrations using special software. The sensitivity of the method was 3 pg/mL and 22 pg/mL for IL-10 and IL-1Ra respectively; the intraseries and interseries error was <10% and the reproducibility index was <99%.
Statistical analysis
The following were used for the statistical analysis of the results: analysis of variance, Student t-test and Pearson correlation coefficient (STATISTICA 6 Pl, StatSoft Inc., Tulsa, OK, USA) at P=0.05.
RESULTS
The IL levels of IL-10 and IL-1Ra from middle ear effusions (or from the tympanic cavity irrigated with 0.5 mL of sterile saline) of 84 children were determined using an ELISA.
IL-1Ra concentrations from the specimens were as follows: group A, 749.85 pg/mL; group NA, 734.16 pg/mL; and group C, 675.07 pg/mL. No statistically significant differences were found in the mean IL-1Ra concentrations between the 3 groups. The results of statistical analyses are shown in Table 2.
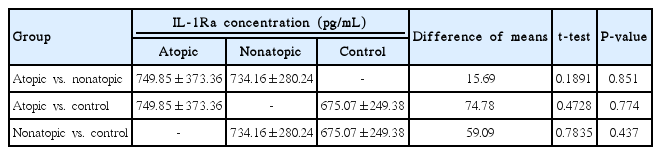
IL-1Ra concentrations (pg/mL) in middle ear effusions from atopic and nonatopic children with COME compared to the control group
The highest mean IL-10 concentration was in effusions from group NA (246.45 pg/mL), followed by group A (229.14 pg/mL). The control group had the lowest mean IL-10 concentration at 193.81 pg/mL. Statistically significant differences (P<0.001) in the mean IL-10 concentrations in specimens collected from the middle ear were found between group NA and group C and between group A and group C. There were no statistically significant differences in the mean IL-10 concentrations between groups NA and A. The results of statistical analyses are shown in Table 3.
DISCUSSION
We investigated the role of two important anti-inflammatory cytokines, IL-1Ra and IL-10, for their role in development of chronic OME. Firstly, we examined the concentrations of IL-1Ra in atopic and nonatopic children with COME compared to a control group. We found no statistically significant differences in the concentrations of IL-1Ra in the specimens collected from the middle ear in children with COME compared to controls. However, the mean concentrations of IL-1Ra in atopic and nonatopic children with COME were slightly higher than in the control group.
Because the levels of IL-1Ra were similar between COME patients and controls, it remains unclear whether IL-1Ra plays a role in disease progression. Indeed, in a recent study using a mouse model of endotoxin-induced middle ear effusions, increased IL-1Ra levels did not appear to alter middle ear effusion production [19]. This indicates that even if IL-1Ra levels are raised during OME, they are not playing an anti-inflammatory role in these patients. However, whether the levels of IL-1Ra were raised to a high enough level in these mice to counteract the pro-inflammatory cytokines is not clear.
The fact that IL-1Ra concentrations in our COME patients were similar to controls could be due to a dysregulation or lack of control over the inflammatory process in the middle ear, regardless of atopy. This dysregulation of the inflammatory immune response, reflected by the similar IL-1Ra levels between the groups, may lead to progression of OME into a chronic state. Thus, increasing the levels of IL-1Ra in patients with OME may help to prevent progression of the disease. Indeed, IL-1Ra administered to mice has been shown to decrease inflammation throughout the course of various disease conditions [10]. More recently, IL-1Ra was administered subcutaneously as a vaccine to patients with rheumatoid arthritis, and was well tolerated and effective in the long-term treatment [20]. Other studies have confirmed the role of IL-1Ra as a modulator in inflammatory conditions, highlighting its potential for application in clinical practice [21].
We also examined the levels of IL-10 in children with COME with and without atopy. As IL-10 down-regulates inflammatory reactions, any imbalance in the production of IL-10 may induce a chronic inflammatory state. In the present study, the mean concentrations of IL-10 were elevated in the specimens from atopic and nonatopic children with COME. This concentration of IL-10 was significantly higher than in the control group. The presence of IL-10 in the effusion could explain the lack of any symptoms of AOM in these patients. However, unfortunately, this means that the damage to the middle ear can occur insidiously and asymptomatically during COME.
The present study did not find any statistically significant differences in the IL-10 concentrations between atopic and nonatopic children with COME, contradicting a previous study where decreased IL-10 levels were observed in atopic patients with allergic asthma [22]. This suggests that IL-10 levels are raised in OME patients regardless of atopy. For the nonatopic patients, the elevated IL-10 levels may be due to the immune response to bacteria found in the middle ear. IL-10 is thought to play a protective role against toxic bacterial polysaccharides. Indeed, in a recent study, a single injection of IL-10 protected mice from a lethal intraperitoneal injection of endotoxin [23].
The elevated levels of IL-10 observed in this study suggest IL-10 may not be capable of effective anti-inflammatory activity, as OME has developed into a chronic condition in these patients. Perhaps an imbalance between the release of IL-10 and the proinflammatory cytokines lead to exacerbation of inflammatory changes in the middle ear cleft. Thus, similar to previous studies, IL-10 may contribute to the shift of an acute condition to COME [24,25]. For example, in experimental studies of AOM, overexpression of IL-10 by T-helper lymphocytes (CD4+) in the middle ear lining was observed [26-28]. This overexpression of IL-10 may downregulate the inflammatory response and could switch the condition from the acute to the chronic state.
It is important to note that it has been hypothesized that the strength of the regulatory IL-10 response reflects the strength of the preceding inflammatory responses [29]. Thus, the levels of IL-10 we observed may not be enough to counteract the strong proinflammatory response observed in the patients prior to our study. We must also consider the fact that although the increased IL-10 levels can switch the condition from the acute to the chronic state, the anti-inflammatory activity of IL-10 may eventually contribute to the resolution of inflammation as observed in patients with allergic asthma [22]. Thus, treatments aimed at increasing IL-10 levels in COME patients may be an effective strategy.
There are already some drugs used for treatment of allergies that result in increased IL-10 production and could be used as treatment in COME [22]. In addition, new immunomodulatory treatments are on the horizon. For example, a study in patients with bronchial asthma showed a new treatment strategy to increase IL-10 levels that is based on a subcutaneously injected immunomodulating glucan [30]. In addition, given that IL-1Ra levels were not elevated in the COME patients in this study, drugs that act to increase levels of this anti-inflammatory cytokine in both atopic and nonatopic patients with COME may also be effective. However, before such drugs are implemented, further investigation of the role of anti-inflammatory cytokines such as IL-10 and IL-1Ra in COME is required.
In conclusion, we observed elevated levels of IL-10 in the effusions of both atopic and nonatopic children with COME. IL-10 can be considered one of the key cytokine mediators, regulating switching of the acute phase of middle ear inflammation into the chronic stage of OME. The results observed in this study may explain the current difficulties encountered in the treatment of these patients and provide arguments for including anti-inflammatory and immunomodulating agents in the management of patients with chronic OME, irrespective of the presence or absence of atopy.
Notes
No potential conflict of interest relevant to this article was reported.